May 26, 2016 — Cancer patients at the Carti Cancer Center, Little Rock Ark., now have access to the latest innovation in diagnostic imaging with Toshiba America Medical Systems Inc.’s Infinix 4-D CT. Carti is the first healthcare provider in the United States to install the Infinix 4-D CT, which merges the Infinix-i angiography system and Aquilion One Vision Edition computed tomography (CT) system. The combination is designed to help clinicians improve visualization and work flow, and increase patient safety.
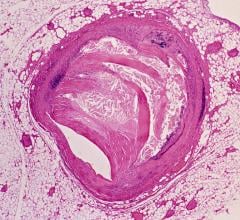
Carti is utilizing the system for interventional radiology and oncology to plan, treat and verify in a single clinical setting. Real CT imaging available on demand may help interventionalists improve visualization of tumors and feeder vessels, as well as provide whole organ body perfusion, to help them not only plan and treat but also verify their procedures. For example, during hepatoma embolizations, interventionalists can immediately visualize and confirm that treatment is hitting the right areas with CT and identify feeder vessels, enabling the interventionalists to guide catheters into the vessels with the angiography system’s 3-D roadmap.
Carti is also using the system during liver ablations and kyphoplasty procedures, among other applications.
“Interventional radiology and oncology is born from innovation, and technology advancements like the Infinix 4-D CT create exciting new possibilities and prepare us for the future of care,” said David Hays, M.D., director, diagnostic and interventional radiology, Carti. “For today’s patients, the system makes procedures that were difficult to perform and turns them into manageable cases that are easier, faster and more accurate.”
The Infinix-i enables clinicians to provide precision and flexibility during intervention with fingertip-to-fingertip and head-to-toe coverage, and enables easier patient access with the Access Halo. The Aquilion One Vision Edition is capable of capturing an entire organ in one rotation with 640 slices with coneXact double slice reconstruction and 16 cm of true anatomical coverage. This produces image quality that far exceeds CT-like imaging of the interventional lab. The combination of the interventional lab and CT eliminates the need to transfer patients between departments and allows clinicians to decrease procedure time and maintain patient safety.
Toshiba’s third-generation iterative dose reconstruction software, AIDR 3-D (Adaptive Iterative Dose Reduction 3-D), offers simplified CT dose reduction, while the Infinix has a suite of dose management features including the Dose Tracking System.
For more information: www.medical.toshiba.com

 October 24, 2025
October 24, 2025