May 23, 2016 — First-in-human data presented at EuroPCR 2016 demonstrate that hemodynamic data from HeartFlow Inc. may help predict which coronary plaques have the potential to rupture. The results were part of a study designed to determine whether use of HeartFlow technology could predict risk of developing acute coronary syndrome (ACS), a condition associated with sudden, reduced blood flow to the heart. The study was led by Professor Bon-Kwon Koo, M.D., Ph.D., FACC, of Seoul National University Hospital.
The EMERALD (Exploring the MEchanism of the plaque Rupture in Acute coronary syndrome using coronary CT angiography and computationaL fluid Dynamics) study evaluated 71 patients who experienced ACS and who had received a coronary computed tomography angiography (cCTA) between one month and two years previously.
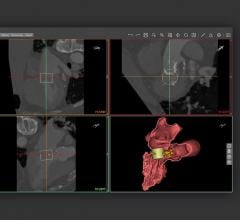
HeartFlow’s hemodynamic assessment, consisting of fractional flow reserve-computed tomography (FFR-CT), and more importantly the change in FFR-CT across the plaque, was a better predictor of which plaques would rupture and lead to ACS than percent diameter stenosis or adverse plaque characteristics (APCs). APCs are features identifiable with cCTA related to plaque composition and disease burden that increase the risk of rupture, as observed in prior clinical studies.
A total of 226 coronary plaques from the cCTAs were assessed – 151 that had not ruptured (non-culprit lesions) and 75 that had ruptured and caused ACS (culprit lesions as identified by invasive angiography). In addition, combining HeartFlow’s hemodynamic assessment and APCs increased the discrimination of lesions at risk for causing ACS more than 10 percent over lesion severity alone.
“When evaluating a patient for risk of ACS, we need to look at not only the burden and composition of the plaque, but also the hemodynamic forces — or stress — placed upon the lesion,” Koo said. “The EMERALD study has demonstrated that by applying this important measurement we may have the potential to greatly improve prediction of the lesions that cause ACS, which could help us optimize treatment strategies for these high-risk patients.”
ACS is a life-threatening condition that hospitalizes approximately 1.4 million Americans each year, of which 810,000 are for myocardial infarction. Studies have shown that plaque rupture causes approximately 75 percent of fatal myocardial infarctions. In the United States alone, an ACS occurs every 25 seconds and someone dies from an ACS-related event every minute.
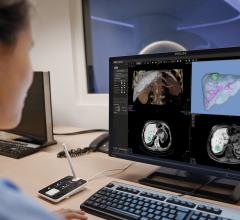
HeartFlow’s initial product, the HeartFlow FFR-CT Analysis, is a non-invasive technology that provides insight into both the extent of coronary artery disease and the impact that disease has on blood flow to the heart, enabling clinicians to select an appropriate treatment. This product is cleared for use in the United States, is also available in Canada, Europe and Japan, and has demonstrated its utility in reducing invasive and costly procedures to diagnose patients suspected of having coronary artery disease.
The hemodynamic factors tested in the EMERALD study include those in the HeartFlow FFR-CT Analysis, which involves the creation of a patient-specific anatomic model from medical imaging data and the application of computational fluid dynamics.
EMERALD is a physician-initiated multicenter, international study conducted at 11 centers in four countries in Europe and Asia. The study evaluated patients who presented with ACS between 2010 and 2014, and underwent a cCTA prior to the ACS event. The goal of the study was to determine whether the evaluation of hemodynamic forces, in combination with adverse plaque characteristics, could improve overall identification of patients at risk for ACS.
For more information: www.heartflow.com

 May 12, 2020
May 12, 2020