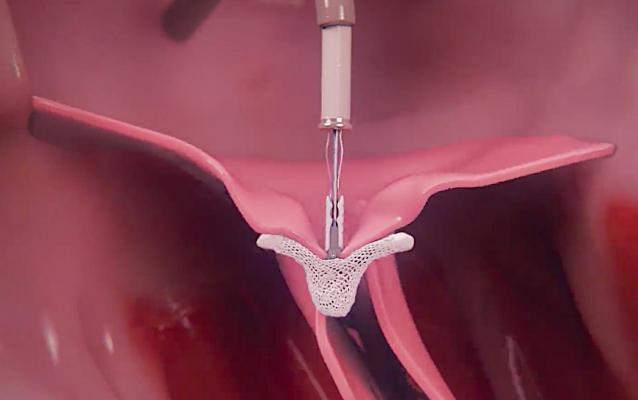
If MitraClip fails and a surgical repair is required, a late-breaking study presented at the 2021 AATS meeting showed 95 percent of patients will require a full surgical valve replacement.
May 4, 2021 – A new study, presented at the 2021 American Association for Thoracic Surgery (AATS) 101st annual meeting, and simultaneously published in the Journal of the American College of Cardiology (JACC), finds that patients suffering from severe mitral regurgitation should be carefully screened and counseled before undergoing transcatheter edge-to-edge mitral repair (TEER).[1] Data showed that up to 95 percent of patients who needed surgery after failed TEER could not have their valves repaired, and needed a full surgical mitral valve replacement to resolve the issue.
This study indicates surgical mitral repair is infrequently achieved after failed TEER, which may have implications for treatment choice in lower-risk and younger patients with degenerative disease. These findings should inform patient consent for TEER, clinical trial design and clinical performance measures, said Joanna Chikwe, M.D., chair of the Department of Cardiac Surgery in the Smidt Heart Institute at Cedars-Sinai and the Irina and George Schaeffer Distinguished Chair in Cardiac Surgery, who presented the results at AATS.
“The fact that TEER takes surgical repair off the table as an option for patients makes it essential that we have a very clear understanding of which patients will benefit. This finding has important implications for treatment choice in lower-risk patients with degenerative disease.” Chikwe said. “These findings should inform patient consent for TEER, design of clinical trials, and clinical performance measures.”
The findings highlight the need to select patients carefully for edge-to-edge mitral repair. The device used for these repairs is the FDA-cleared Abbott MitraClip. The device is delivered through a catheter under transesophageal echo (TEE)image guidance to grasp both valve leaflets and clip them in place, mimicking a surgical repair to reduce or eliminate regurgitant flow. The MitraClip uses two metal arms and tiny barbs to secure the device to the leaflets and the device becomes endothelialized as a permanent part of the valve leaflet structure.
As a less invasive approach, TEER is an attractive alternative treatment to mitral valve surgery, particularly in older patients. However, TEER is not currently recommended for younger patients who benefit more from a surgical repair long-term. This caution is supported by this study of 463 patients which provides the first national data on surgical outcomes when TEER fails. The surgical repair rate after failed TEER was only 4.8 percent overall, and 6.8 percent in degenerative disease.
Study Data Details
In the study cohort of 463 patients, the median age was 76 years, median left ventricular ejection fraction 57% (IQR 48-62%), and 177 (38.2%) patients had degenerative disease. Major concomitant cardiac surgery was performed in 137 (29.4%) patients: in patients undergoing isolated mitral surgery median STS predicted mortality was 6.5% (IQR 3.9-10.5%), observed mortality was 10.2% (n=23/225) and the ratio of observed to expected mortality was 1.2 (95% CI 0.8-1.9).
Predictors of mortality included urgent surgery (odds ratio (OR) 2.4, 95% confidence interval (CI) 1.3-4.6), non-degenerative / unknown etiology (OR 2.2, 95% CI 1.1-4.5), creatinine >2.0mg/dl (OR 3.8, 95% CI 1.9-7.9) and age >80 (OR 2.1, 95% CI 1.1-4.4).
In a volume outcomes analysis in an expanded cohort of 591 patients at 227 hospitals, operative mortality was 2.6% (n=2/76) in 4 centers that performed >10 cases versus 12.4% (n= 64/515) in centers performing fewer (p=0.01). The surgical repair rate after failed TEER was 4.8% (n=22), and 6.8% (n=12) in degenerative disease.
Results of additional industry studies and an NIH-funded trial comparing TEER to mitral surgery should be published within the next two years and may result in updated guidelines on the use of TEER to treat degenerative mitral regurgitation.
The AATS is the premiere association for cardiothoracic surgeons in the world and works to continually enhance the ability of cardiothoracic surgeons to provide the highest quality of patient care. It is more than 1,500 members have a proven record of distinction within the specialty and have made significant contributions to the care and treatment of cardiothoracic disease.
For more information: www.aats.org
Find more heart valve technology news
Reference:


 May 06, 2026
May 06, 2026 









