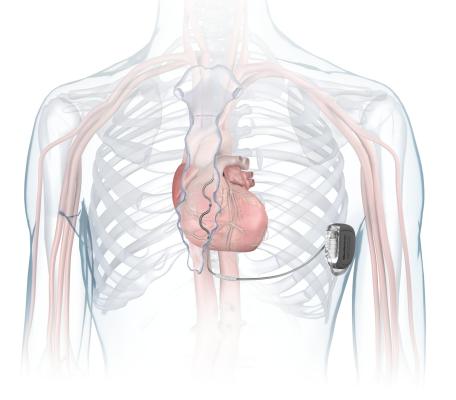
The Aurora EV-ICD system
October 23, 2023 — Medtronic plc, a global leader in healthcare technology, has received U.S. Food and Drug Administration (FDA) approval for the Aurora EV-ICD MRI SureScan (Extravascular Implantable Cardioverter-Defibrillator) and Epsila EV MRI SureScan defibrillation lead to treat dangerously fast heart rhythms that can lead to sudden cardiac arrest (SCA). The Aurora EV-ICD system is the first-of-its-kind to provide the life-saving benefits of traditional, transvenous ICDs with a lead (thin wire) placed under the breastbone, outside of the heart and veins. The Aurora EV-ICD delivers lifesaving defibrillation, anti-tachycardia pacing (ATP), and back-up (pause-prevention) pacing therapies via a device similar in size, shape, and longevity to traditional, transvenous ICDs.
eastbone (sternum) using a minimally invasive approach. The Epsila EV defibrillation lead is placed outside of the heart and veins, helping to avoid certain complications associated with transvenous leads, such as vascular injury and vessel occlusion (narrowing, blockage or compression of a vein).
The Aurora EV-ICD system includes features available in Medtronic transvenous ICDs, and offers additional advantages that are not available with the subcutaneous ICD including:
- Anti-tachycardia Pacing (ATP), to terminate ventricular arrhythmias (rapid and/or chaotic activity of the heart that can lead to SCA) using low-energy pacing pulses, potentially avoiding a defibrillation shock.
- Pause Prevention Pacing, to provide back-up pacing for brief, intermittent, heartbeat pauses.
- 40 Joule Defibrillation Energy, to deliver life-saving shocks in a device the size of transvenous ICDs (33 cc)
- Medtronic exclusive PhysioCurve design, to increase patient comfort and implant acceptance.
- 11.7-year projected longevity, to reduce device replacement procedures during a patient's lifetime.
Patients who receive the commercially available Aurora device also will benefit from the addition of Smart Sense, a proprietary algorithm designed to reduce the potential for inappropriate shocks.
Medtronic will obtain real-world performance and safety data on the Aurora system in the Enlighten global post-approval registry, a prospective, non-randomized, observational, multicenter study, expected to last five years and enroll approximately 1,000 patients. First implants in Enlighten and first commercial implants worldwide were recently conducted by Lucas V.A. Boersma, M.D., Ph.D., cardiologist at St. Antonius Hospital, Nieuwegein, The Netherlands, and a limited launch is underway in select European countries.
The Aurora EV-ICD system is indicated for patients who are at risk of life-threatening arrhythmias, and who have not had a prior sternotomy and do not need chronic bradycardia (abnormally slow heartbeat) pacing.
About the EV ICD Pivotal Study
The EV ICD Pivotal study is a prospective, multicenter, single-arm, non-randomized, pre-market clinical study that assessed the safety and effectiveness of the Medtronic EV ICD system for patients at risk of sudden cardiac death. It enrolled 356 patients at 46 sites in 17 countries in North America, Europe, the Middle East, Asia, Australia and New Zealand, and results were published in The New England Journal of Medicine. Safety and effectiveness results were sustained out to 18 months.2
For more information: www.medtronic.com
References:
1Friedman P, Murgatroyd F, Boersma LVA, et al. Efficacy and Safety of an Extravascular Implantable Cardioverter-Defibrillator. N Engl J Med 2022; 387:1292-1302.
2Friedman P, Murgatroyd F, Boersma LVA, et al. Chronic Safety and Performance of the Extravascular ICD: Results from the Global EV ICD Pivotal Study. Heart Rhythm Society late breaking clinical trials, May 20, 2023.


 April 20, 2026
April 20, 2026 









