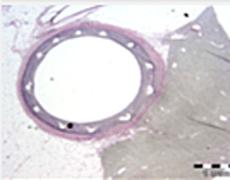
June 30, 2011 — Micell Technologies Inc. announced that it has completed its review of the scheduled four-month follow-up on the first 10 patients from the DESSOLVE I first-in-human trial of the MiStent drug-eluting coronary stent system ("MiStent DES"), an ultra-thin drug-eluting stent (DES) distinguished by a rapid-absorbing drug/polymer coating formulation. Based on results observed in the DESSOLVE I trial, Micell has reduced the sample size in its DESSOLVE II CE Mark study from 270 to 171 planned subjects.
DESSOLVE I, the first-in-human study of the MiStent DES comprising 30 patients with documented stable or unstable angina pectoris, completed enrollment earlier this year. The primary endpoint is in-stent late lumen loss, as measured by the angiography core laboratory in de novo lesions ranging in diameter from 2.5 to 3.5 mm and amenable to treatment with a maximum 23 mm length stent.
Dennis J. Donohoe, M.D., Micell's chief medical advisor, said, "Encouraging results in minimizing late lumen loss with the MiStent DES in the DESSOLVE I trial has prompted us to reduce the total sample size of our pivotal DESSOLVE II trial, with the full agreement of Micell's clinical advisors and principal investigators following rigorous data evaluation."
The DESSOLVE II CE Mark trial is an ongoing multi-center study of patients with documented stable or unstable angina pectoris. The primary endpoint is superiority of the MiStent DES in minimizing in-stent late lumen loss at nine months, compared to Medtronic's Endeavor Sprint DES, as measured by the angiography core laboratory in de novo lesions ranging in diameter from 2.5 to 3.5 mm and amenable to treatment with a maximum 30 mm length stent.
For more information: www.micell.com


 May 06, 2026
May 06, 2026 









