
March 14, 2018 — Breast cancer patients who started taking one of two well-known heart medications at the same time they initiated trastuzumab — a targeted cancer therapy that has been linked to heart damage — received no benefit in terms of preventing declines in heart function, according to research presented at the American College of Cardiology’s 67th Annual Scientific Session. However, in patients who had received or were concurrently receiving anthracycline-based chemotherapy in addition to trastuzumab, the occurrence of heart damage was halved among those taking either the angiotensin converting enzyme inhibitor (ACE-inhibitor) lisinopril or beta blocker carvedilol, compared with placebo after two years of follow-up.
“Our findings suggest that among women who are only on a standard course of trastuzumab neither carvedilol nor lisinopril seem to make a difference, but for those who had a history of being on anthracycline, these medications can be cardioprotective and should be considered,” said Maya E. Guglin, M.D., professor of medicine in the Division of Cardiovascular Disease, University of Kentucky, and lead author of the study.
This study comes amid growing awareness that certain cancer therapies can contribute to heart failure or other heart problems. While smaller studies have looked at the utility of these commonly-used heart medications in guarding against anthracycline-induced cardiotoxicity, this trial is one of the first and the largest to date to test whether these drugs can prevent trastuzumab-related heart damage and prevent the need to discontinue a potentially life-saving treatment.
“We wanted to see if the cardiopreventive effects apply to trastuzumab,” Guglin said.
Trastuzumab (Herceptin) has been lauded as a breakthrough treatment for HER2-positive breast cancer, an especially aggressive type of cancer. However, Guglin said some studies have shown that up to 1 in 4 women who receive this therapy experience cardiac effects, including symptomatic heart failure, a condition in which the heart becomes too weak to pump enough blood to meet the body’s needs. This has led to stricter clinical guidelines requiring women to be screened via echocardiogram or other imaging modality before starting trastuzumab to assess their ejection fraction — the percent of blood volume that is ejected from the left ventricle, the main pumping chamber of the heart, with each heartbeat. If the ejection fraction is less than 50 percent (low limit of normal), they are not eligible for trastuzumab based on current practice standards.
“It’s a very difficult scenario, because we don’t want to damage their heart, but at the same time we certainly don’t want to compromise the chances of [a] cure from cancer,” she said. “Because of this initial screening process, we deny [some of] them potentially life-saving medication.”
Guglin explained that while anthracycline chemotherapy can cause long-lasting, potentially irreversible damage to the heart muscle, trastuzumab therapy has been associated with milder and often temporary declines in cardiac function.
“Our data affirm that anthracyclines are aggressive agents that can cause damage to the heart muscle on a much greater scale than trastuzumab,” she said, adding that the damage may already be done from prior exposure to this type of chemotherapy. So, while their ejection fraction is normal, making them eligible to start on trastuzumab, their heart may already be compromised.
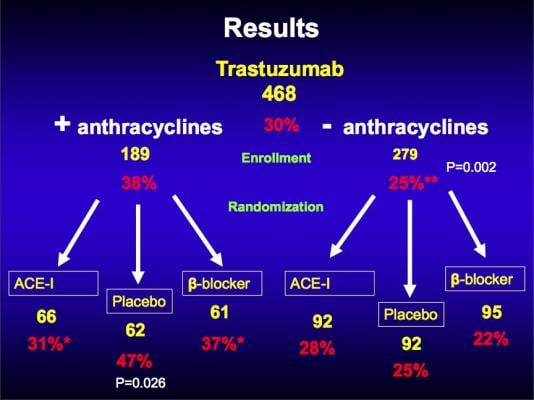
This prospective, randomized, controlled trial enrolled 468 patients at more than 165 centers in North America who were diagnosed with breast cancer and for whom trastuzumab was clinically indicated. All study participants had normal heart function measured by ejection fraction and were not already taking an ACE-inhibitor or beta blocker for other medical reasons. Half were receiving or had received anthracycline-based chemotherapy. Patients were randomly assigned to receive once daily lisinopril (10 mg), carvedilol (10 mg Coreg CR) or placebo and were followed for two years (the year of active treatment with trastuzumab and the year that followed). The groups were comparable in terms of age (average of 51 years) and cardiovascular risk factors. Researchers assessed heart function and any declines with echocardiograms every three months.
Neither lisinopril nor carvedilol were statistically different from placebo in terms of preventing cardiotoxicity caused by trastuzumab or preventing related disruptions in therapy. However, in patients who were treated with anthracyclines, these heart medications were effective in preserving left ventricular ejection fraction. Carvedilol and lisinopril reduced declines in heart function by half, which was a statistically significant difference (odds ratio of 0.49 and 0.53, respectively, compared with placebo). Adverse cardiac effects were similar across the three groups, occurring in 32 percent of patients on placebo, 29 percent of those on carvedilol and 30 percent on lisinopril. Common adverse events were low blood pressure and dizziness, which were milder in the carvedilol group.
“Our study indicates that carvedilol is tolerated better,” Guglin said. “But based on our study, if you have breast cancer and your oncologist wants to start you on Herceptin and you’ve been on an anthracycline, you have a better chance of avoiding decline in cardiac function and taking Herceptin without damaging your heart if you are pretreated with lisinopril or carvedilol, whichever is tolerated better.”
She is quick to stress that not all cancer treatment therapies cause heart problems and, for those that do, there may be ways to potentially minimize the risk of related heart damage.
Pamela Munster, M.D., an oncologist and senior study author said this trial gives important data for clinicians to use when planning cancer treatments for high-risk patients. “This study is of great importance as it provides the oncologist the option to use an anthracycline in HER2 positive, early-stage breast cancer patients,” she said. “Anthracycline-based regimens are more effective for high-risk patients, but the increased cardiotoxicity has limited its use.”
The researchers will continue to analyze the data and evaluate how taking heart medications alongside targeted cancer therapy affects the magnitude of cardiac dysfunction, how long it lasted and whether or not it can be reversed.
The study was co-sponsored by the University of South Florida and the National Cancer Institute.
For more information: www.acc.org
Related Content
Beta Blocker Shows Mixed Results in Protecting Against Chemo-Induced Heart Damage
AHA Puts New Focus on Where Heart Disease and Breast Cancer Treatment Meet
VIDEO: Linking Breast Cancer Treatment to Cardiac Issues
VIDEO: Assessing Cardiotoxicity Response With Cardio-Oncology Echo Imaging


 April 02, 2026
April 02, 2026 









