April 1, 2020 — Three months after undergoing renal denervation (RDN), patients with untreated high blood pressure had statistically significant reductions in average blood pressure over 24 hours compared with patients who underwent a sham procedure and experienced no major adverse effects, according to results from the SPYRAL-HTN OFF MED pivotal trial presented at the American College of Cardiology (ACC) 2020 Annual Scientific Session. The study tested the efficacy and the Medtronic Spyral catheter based RDN system.
“These results show that RDN offers an effective alternative approach to traditional medications that require patient adherence for reducing blood pressure,” said Michael Böhm, M.D., of Saarland University Medical Center in Hamburg, Germany and lead author of the study. “Furthermore, the findings show that RDN lowers blood pressure not just during the day but also throughout the night and early-morning periods when risk is highest for [adverse] clinical events and the effect of some medications on blood pressure is reduced.”
High blood pressure, also known as hypertension, is a leading cause of heart attacks, strokes and death. It is estimated that 46 percent of adults in the U.S. have high blood pressure based on the 2017 ACC/AHA High Blood Pressure in Adults guideline.
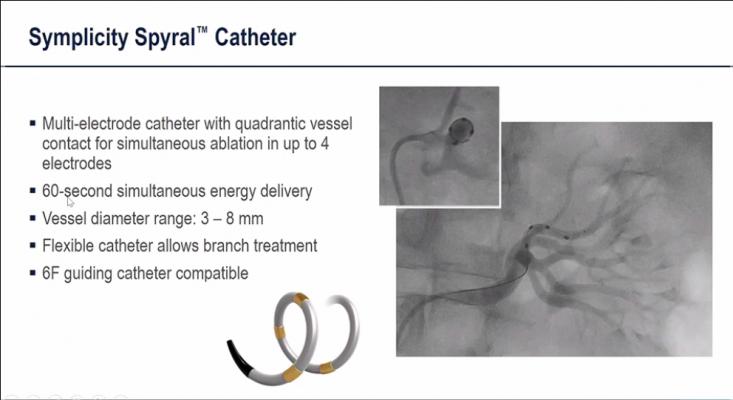
During renal denervation procedures, energy is delivered to overactive nerves in the renal arteries between the aorta and the kidneys, which eliminates vasodilation and props the arteries open to allow more blood filtration by the kidneys remote access water an help reduce hypertension.

Michael Böhm, M.D., said there were significant reductions in average blood pressure using the Medtronic Spiral renal denervation system, according to the data presented at ACC 2020 from the SPYRAL-HTN OFF MED pivotal trial.
Blood pressure-reducing medications are often insufficiently effective, Böhm said. Earlier research has shown that among adults in the U.S. who take high blood pressure medication, approximately half still have elevated blood pressure. In addition, some people with high blood pressure either do not want to take medication or cannot take it due to intolerable side effects, he said. For these reasons, effective non-drug treatments for high blood pressure are needed.
The kidneys help to regulate blood pressure by removing salt and excess water from the body. However, over activity of the sympathetic nerves in the arteries leading to the kidneys can contribute to high blood pressure. RDN works by delivering an electrical current that decreases the overactivity of these nerves. Pilot studies have shown that, compared with a sham procedure, RDN reduces blood pressure.
RDN is performed in a hospital catheterization lab (cath lab) under local anesthesia and with imaging guidance. A catheter is threaded from the femoral artery in the thigh to the arteries that serve the kidneys. There, the catheter delivers pulses of radiofrequency energy to the nerves in the arteries. Patients typically spend one night in the hospital after the procedure.
The SPYRAL-HTN OFF MED pivotal trial was designed in collaboration with the U.S. Food and Drug Administration (FDA) to provide statistically significant evidence of whether RDN could reduce blood pressure in patients not taking blood pressure-reducing medication.
The study, which was conducted at 46 sites in nine countries (the U.S., Germany, Greece, Australia, Canada, Japan, the United Kingdom, Austria and Ireland), enrolled 331 patients whose average systolic blood pressure (SBP; the top number) over 24 hours was between 140 and 170 mm Hg. Their average age was 53 years, and 67 percent were men. Most had obesity, with an average body mass index of 31. Around 5 percent also had Type 2 diabetes. A condition of enrollment was that patients either had not been taking any blood pressure-reducing medications or had discontinued those medications at least three weeks before their blood pressure was measured at study entry.
Patients were randomly assigned to receive either RDN or a sham procedure in which a catheter was inserted and only an angiography (X-ray of the blood vessels) was performed. A total of 166 patients were assigned to RDN and 165 to the sham procedure. Patients did not know whether they were receiving RDN or the sham procedure. The doctors and nurses who measured patients’ blood pressure and treated them after the procedure also did not know which group patients were in.
At the end of three months, the primary efficacy endpoint was the change in average 24-hour SBP, adjusted for SBP at study entry. The secondary efficacy endpoint was the change in average blood pressure measured in the doctor’s office at three months, adjusted for office blood pressure at study entry. Major adverse safety events were also assessed at three months, including rates of death, stroke, changes in kidney function or any injury to the arteries surrounding the kidney.
For the primary and secondary efficacy endpoints, the difference between groups was –3.9 mm Hg for 24-hour SBP and –6.5 mm Hg for office SBP, both favoring the RDN group, with a 99.9 percent probability that RDN was superior to the sham procedure.
No deaths, strokes or changes in kidney function occurred during the three-month follow-up period, Böhm said. He added that these results may not demonstrate the total decrease in blood pressure achieved with RDN given the short follow-up period due to ethical and safety concerns that patients needed to be restarted on antihypertensive medications. In other studies of RDN, however, patients’ blood pressure has continued to decline at six months or more after the procedure, he said.
“This study establishes RDN as an additional option beyond exercise or lifestyle modification for patients with high blood pressure who are unwilling to take or cannot tolerate medication,” Böhm said, adding that the study results will be included in an application for FDA approval of the RDN device used in the trial for the treatment of patients who are not taking blood pressure-reducing medication. Currently there are no RDN devices approved for use in the U.S.
Results from a companion study, the SPYRAL-HTN ON MED trial, which is testing the safety and effectiveness of RDN in patients who are taking up to three blood pressure-reducing medications, are expected in about 18 months. The results of that study will also be included in the application for FDA approval of the RDN device, Böhm said.
This study was funded by Medtronic Inc.


 April 02, 2026
April 02, 2026 









