June 2, 2009 - Transoma Medical Inc., manufacturer of Sleuth and Sleuth AT wireless, automated implantable cardiac monitoring (ICM) systems, received FDA 510(k) marketing clearance for its new data review feature, allowing clinicians to easily access up to 99 events stored on the patient’s Personal Diagnostic Manager (PDM).
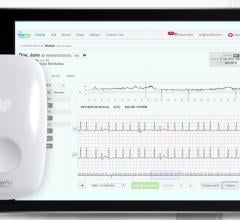
The PDM is a handheld device that retrieves and stores ECG data from the implanted device prior to automatic transmission to the 24/7 monitoring center. Each stored ECG strip is labeled with the time and date of the cardiac event making it easier for clinicians to correlate symptoms with rhythms.
“Instant access to a patient’s ECG data on the mobile handheld device is a clinically valuable tool in urgent situations, such as when the patient presents in the emergency room,” said Asim Yunus, M.D., an electrophysiologist with Michigan Cardiovascular Institute in Saginaw, Mich. “Transoma continues to advance cardiac monitoring technology with their devices. The Sleuth systems already provided information much faster and more efficiently than other implantable monitoring options. Now, with Data Review, we don’t even have to wait for the automatic downloads to the monitoring center because the events are right there on the handheld device. This kind of access to information has never been available before.”
As a complete, remote and automated system, Sleuth AT offers several advantages over other available implantable monitoring products, the company said. The system includes a small implanted device that incorporates the most sensitive arrhythmia detector of any ICM on the market. It captures ECG strips containing arrhythmias automatically and allows the patient to capture data by pressing a button on the handheld PDM after experiencing a symptom. All data captured by the implanted device is automatically transferred to the PDM, providing an additional 10.5 hours of ECG storage. Data stored on the PDM is automatically transmitted to a 24/7 monitoring center where certified cardiac technicians review the data and alert clinicians to clinically significant events, providing a high degree of specificity. And now, with data review, ECG recordings that have not yet been transmitted to the monitoring center can be accessed directly on the handheld PDM.
“As a device clinic coordinator, it is not uncommon for me to manage data on 2,000 or more patients at a given time from a variety of cardiac monitoring systems,” said Lori Fravel, BSN, CCRN, CCDS, a nurse who supports 10 cardiologists at Mt. Carmel Columbus Cardiology Consultants in Columbus, Ohio. “I have found the Sleuth systems to be extremely efficient because I am only alerted to clinically-significant events and I never have to call the patient to coordinate manual interrogation of his or her device. The setup of the system allows me to get the information I need in a much more streamlined fashion. Now with Sleuth AT, the added benefit of ECG access on the handheld device means any available clinician can view recently captured events without a programmer, even if the patient has been away from home. I am hopeful this will further speed access to critical information.”
The Sleuth AT is a small, thin, implantable cardiac monitor about the size of a 50-cent piece (or the size of the smallest pacemakers), which continuously monitors the electrical activity of the heart. The cardiac monitor is implanted under the skin during a brief, outpatient procedure.
The Personal Diagnostic Manager (PDM) is a handheld, multipurpose device that automatically retrieves and stores relevant ECG data from the implanted cardiac monitor and securely relays the information through a base station to the monitoring center. The PDM is also used by the physician to program the implanted cardiac monitor and to view nontransmitted ECGs immediately after capture. Data are collected in three ways:
For more information: www.transoma.com


 July 22, 2025
July 22, 2025