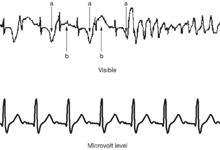
The top line shows the visible ECG, and the bottom shows the microvolt T-waves.
November 13, 2009 – Microvolt T-Wave Alternans (MTWA) technology from Cambridge Heart Inc. will be incorporated into a new module being developed for Cardiac Science’s cardiac stress test systems.
The MTWA technology integration will be distributed to cardiologists and primary care physicians and will make it more accessible. Cambridge Heart estimates 10 to 12 million higher-risk cardiac patients can benefit from an annual MTWA test, which helps detect patients with early symptoms for sudden cardiac death.
Cambridge Heart has completed the prototype development of the module, which is now subject to industry standard validation and verification processes, and regulatory approval.
For more information: www.cardiacscience.com


 May 08, 2026
May 08, 2026 









