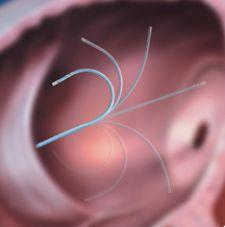
A new ablation catheter designed to give physicians improved range of motion and ease of use during procedures to eliminate abnormal heart rhythms, the EZ Steer Bi-Directional Catheter helps physicians restore atrial tachycardias into normal sinus rhythm. This enables the heart to efficiently pump blood to the body's vital organs. The defining feature of this product is its micro-tensioning system, which allows physicians — through a series of very small, continuous micro-movements — to progressively dial in the precise force desired for deflection of the catheter. Because the micro-tensioning system can hold the deflection in place, there is no need for repeated locking and unlocking of the curve position. Physicians experience smoother deflection and greater predictability. To better diagnose and treat certain arrhythmias, the EZ Steer Catheter incorporates an ergonomically-inspired handle, a design that aids comfort and provides the physician with a feeling of "direct connection" with the catheter tip and target ablation area.


 February 12, 2026
February 12, 2026 









