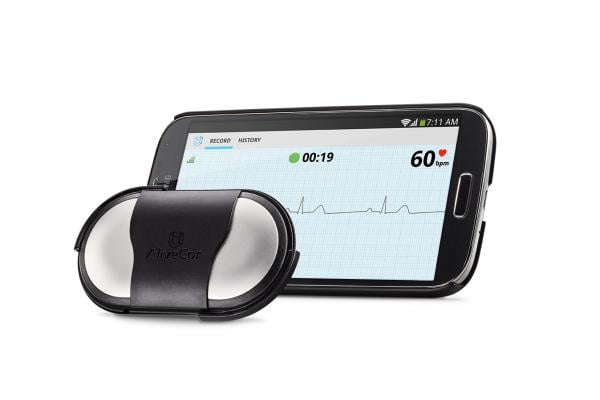
October 7, 2013 — At the University of Southern California Body Computing Conference, AliveCor Inc. announced its new versatile Heart Monitor, a device that attaches to compatible smartphones and smartphone cases.
The new design includes the proprietary AliveCor ultrasound technology that enables a clinical-quality electrocardiogram (ECG) recording of the heart’s activity while minimizing battery drain. The compact design is transferable among various mobile devices.
The U.S. Food and Drug Administration (FDA)-cleared Heart Monitor attaches directly to the back of the mobile device or its case with the universal attachment plate. Current Android devices supported are the Samsung Galaxy S3, S4, and HTC One. Apple devices supported are the iPhone 4, 4S, 5 and the iPod touch 5. AliveCor also offers cases for the Galaxy S4 and for the iPhone 4, 4S, and 5.
For more information: www.alivecor.com


 May 08, 2026
May 08, 2026 









