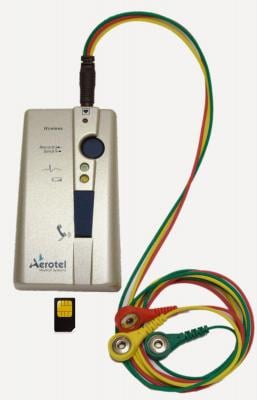
March 25, 2014 — Aerotel Medical Systems has received the CE Mark approval for its new electrocardiogram (ECG) devices, HeartView P12/8i and HeartView 12L.
The HeartView 12L, a high-resolution 12-lead ECG recorder and transmitter for professional use is one of the world's smallest and most powerful ECG monitors. The new HeartView P12/8i, an ECG event recorder-transmitter for personal use, is a powerful ECG device with high-resolution 12-lead ECG recording technology, specifically designed for patient use.
Both devices enable the transmission of comprehensive ECG data from any place at any time. The recorded ECG is transmitted automatically via Bluetooth to Aerotel's Mobile CliniQ App. The ECG data is then automatically sent from the mobile device to a cardiac call center for immediate diagnosis.
The HeartView devices allow:
- Diagnosis of cardiac symptoms
- Remote monitoring of chronic heart patients
- Recording and transmission of an ECG to a cardiac call center
- Cardiac analysis for clinical studies
For more information: www.aerotel.com


 March 06, 2026
March 06, 2026 









