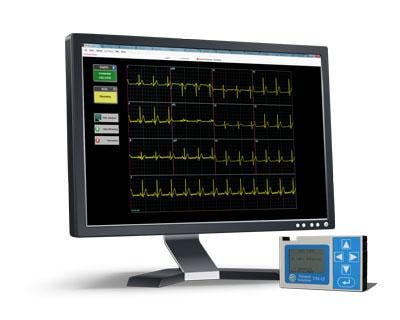
June 30, 2014 — Telemed Solutions has reached an agreement with GlobalMedia Group LLC to become the exclusive global distributor for the TM-12, a PC-based wireless resting 12-lead ECG. GlobalMed also will act as the exclusive master distributor for order fulfillment to Telemed Solutions existing distributors, resellers and customers.
Telemed Solutions CEO Ken Burns expects this relationship to get the TM-12 in markets and countries much sooner than once realized. “GlobalMed has relationships and experience in the private sector in addition to Federal and State agencies both domestic and abroad that will get the TM-12 into areas that would have taken us much longer to do so. In turn, we will be able to put more focus on our ECG Management System and concentrate on our other products,” Burns said.
GlobalMed will combine its expertise in hardware/software integration, international sales and marketing, product distribution and customer service with Telemed Solutions’s technology and expertise in ECG and related software to achieve a combined sales and marketing effort and hardware/software integration platform that will significantly broaden the revenue potential for both companies.
With the agreement, GlobalMed will use their extensive relationships and experience in the healthcare agencies of the U.S. Federal and state governments. GlobalMed shall offer Telemed Solutions products as pre-engineered, made in the United States, easily integrated retrofit additions to GlobalMed’s existing examination station footprint in the U.S. Department of Veteran’s Affairs and to the other Federal and State agencies currently utilizing GlobalMed’s examination stations and software. GlobalMed will rebrand the CardioVu software to TotalECG.
For more information: www.telemedcorp.com


 May 08, 2026
May 08, 2026 









