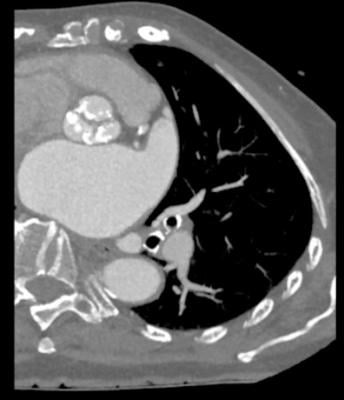
May 7, 2018 — Colibri Heart Valve LLC announced the Colibri transcatheter aortic valve implantation (TAVI) System has been used on an additional four patients in the ongoing international, single-arm, open-label early feasibility study (EFS). Initial post-implantation results from these patients, who received a 24mm valve, show favorable low aortic valve pressure gradients and no observed paravalvular leakage or aortic insufficiency. The Colibri TAVI System is being developed with 21-, 24-, 27- and 30mm valves to accommodate a variety of clinical and patient needs.
Thirty-day follow-up results from these patients, as well as from those implanted with the Colibri 27mm valve in March, will be available at the Colibri Heart Valve Exhibit booth at the EuroPCR 2018, May 22-25 in Paris, France.
The ongoing EFS will enroll up to 10 individuals with severe aortic stenosis, who will be implanted with Colibri’s second-generation 24- or 27mm percutaneous aortic heart valve in a TAVI procedure. This proprietary TAVI system features a replacement heart valve pre-mounted and pre-crimped on a balloon delivery catheter, pre-loaded into a low-profile access sheath and sterilized, ready-for-use from package to patient.
Colibri President and CEO Joseph B. Horn added that the 27mm valve, on average, with the Colibri folded leaflet technology, demonstrated an aortic valve area of 3 cm2 and a single-digit valve gradient. He said that this represents a significant improvement in clinical performance in the heart valve marketplace, and that it will alleviate the patient prosthetic mismatch (PPM) that is observed with currently available heart valves.
For more information: www.colibrihv.com


 May 06, 2026
May 06, 2026 









