June 29, 2023 — UltraSight, a digital health pioneer transforming cardiac imaging through the power of artificial intelligence, announced a partnership with point-of-care ultrasound innovator EchoNous, to enable more healthcare professionals to perform cardiac ultrasound and help increase patient access to cardiac care. The new collaboration was announced at ASE 2023, the premier event for bringing together echo professionals.
“Empowering a greater number of healthcare professionals in more care settings to conduct cardiac ultrasound brings us one step closer to realizing our vision of making cardiac care more equitable for patients around the world,” said UltraSight CEO Davidi Vortman. “We know that image quality is the key for achieving accurate patient diagnosis. This is why we are thrilled to collaborate with EchoNous, which provides superior image quality and offers one of the most advanced handheld ultrasound systems available on the market.”
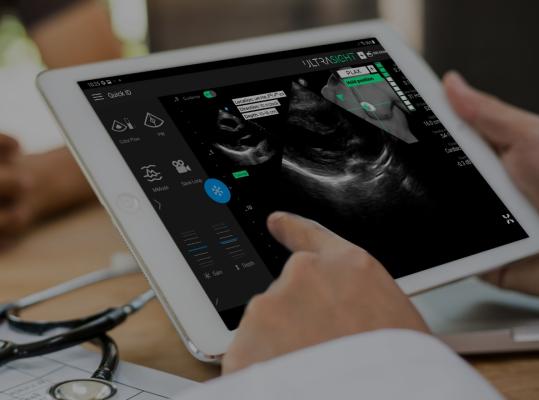
UltraSight’s real-time AI guidance software will be integrated with EchoNous’ KOSMOS handheld ultrasound device with the goal of enabling chronic cardiac patient monitoring to be done closer to patients. Fusing together these joint capabilities, the goal is for healthcare professionals with no prior sonography training to be able to accurately conduct echocardiographic examinations.
UltraSight will present its pivotal study results and EchoNous will be exhibiting at the American Society of Echocardiography’s 34th Annual Scientific Sessions held on June 23-26, 2023 in National Harbor, Maryland. UltraSight’s study, which is the basis for the company’s FDA submission, demonstrates that novice users with real-time AI guidance software were able to successfully acquire diagnostic quality cardiac ultrasound images.
“UltraSight's AI solution in conjunction with our own AI technology will help widespread use and adoption to provide Echocardiology exams in any location or setting,” said Graham Cox, CEO of EchoNous. “With this partnership, we will work toward offering remote clinicians the tools they need to make lifesaving diagnoses, cardiac monitoring and potentially improve cardiac care for patients in rural areas.”
EchoNous released the groundbreaking Kosmos handheld ultrasound device in 2020 and has continued refining the platform since then. Available on iOS, Android and its own proprietary system, the AI-enhanced Kosmos ultrasound system boasts capabilities and image quality that is comparable to cart-based systems at a lower cost. A partnership with UltraSight can provide complementary AI guidance that further enhances Kosmos’ AI offering.
“Providing more professionals in the medical community with the ability to take high-quality diagnostic images of the heart has the potential to yield enormous clinical benefits,” said Dr. Peter J. Fitzgerald, MD, PhD, at Stanford University. “Not only do we have a chance to close the gap between novice and skilled ultrasound users, but this new technology may help alleviate the bottleneck related to sonographer shortages that continues to plague today’s healthcare system.”
When paired with the EchoNous Kosmos, UltraSight’s underlying AI neural network predicts the position of the ultrasound probe relative to the heart based on the ultrasound video stream and guides the user on maneuvering the probe to capture diagnostic quality cardiac images.
After acquiring diagnostic-quality cardiac images at the point of care, images can then be read remotely. By separating the acquisition and interpretation of ultrasound images, UltraSight technology has the potential to improve access to optimized cardiac care and more cost-effective heart monitoring opportunities. The UltraSight real-time AI guidance software is currently under FDA 510(k) review.
For more information: www.ultrasight.com, www.EchoNous.com.


 May 12, 2026
May 12, 2026 









