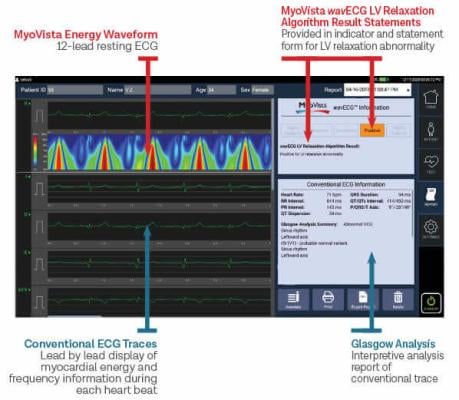
August 24, 2023 —Heart Test Laboratories, Inc. d/b/a HeartSciences, an artificial intelligence (AI)-based medical technology company focused on transforming ECGs/EKGs to save lives through earlier detection of heart disease, announced that its MyoVista wavECG has been selected by PanaceaFlo Ltd to be used in Dublin, Ireland in its heart screening evaluation study.
The main objective of the study is to establish a cardiovascular risk screening program and integrated care hub pathway. If successful, the program is expected to roll out nationally in Ireland, in conjunction with the Irish Health Elevator Program, focusing on community-based screening and early intervention of cardiovascular risk in line with the Irish public health system (HSE) “Stayleft, Shiftleft10X” transformation program to move to preventative rather than acute care.
It will initially screen approximately 250 members of the general public, including Garda Siochana officers, in Dublin, Ireland, in collaboration with the Innovation Value Institute (IVI), Maynooth University College, Kildare.
Andrew Simpson, CEO of HeartSciences, stated, “We are delighted that the MyoVista wavECG has been chosen as part of a major frontline heart screening evaluation program. It further validates the enormous commercial opportunity for the MyoVista and the role of AI ECG. Heart disease is placing considerable strain on health systems worldwide and is the largest health and cost burden. I believe that preventative testing using AI ECG will be the key to saving lives and reducing cardiology outpatient backlogs, waiting list times and system costs. Our ultimate objective is for the MyoVista to become a fixture in frontline healthcare to help physicians make better and earlier referral decisions for at risk patients."
For more information: www.heartsciences.com


 May 08, 2026
May 08, 2026 









