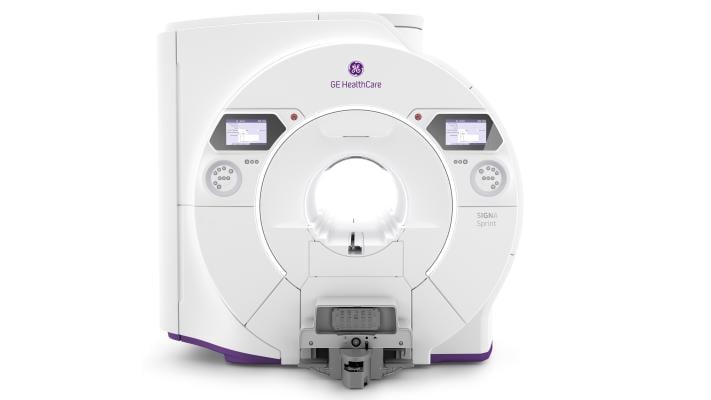
May 12, 2025 — GE HealthCare recently unveiled Signa Sprint, an FDA 510(k) pending[1] ultra-premium wide bore 1.5T high-performance gradient MRI system, at the International Society for Magnetic Resonance in Medicine (ISMRM) 2025. This technology is designed to unlock advanced imaging possibilities in cardiology, oncology and other clinical and research areas.
The need for advanced diagnostics in cardiology and oncology continues to grow, with cardiovascular disease accounting for 32% of global deaths,[2] and cancer accounting for nearly 10 million deaths per year.[3] Meeting this demand with advanced MRI technologies has the potential to make significant impact for cardiology and oncology patients.
With the goal of delivering higher gradient benefits previously attainable on only 3.0T systems, Signa Sprint is designed to offer high-performance scanning made simple at 1.5T, with exceptional diffusion imaging, a critical tool in oncology diagnosis and treatment planning. Signa Sprint is also designed to equip clinicians and researchers with the ability to expand the current 1.5T boundaries, aiming to enable fast clinical translation and pioneering research.
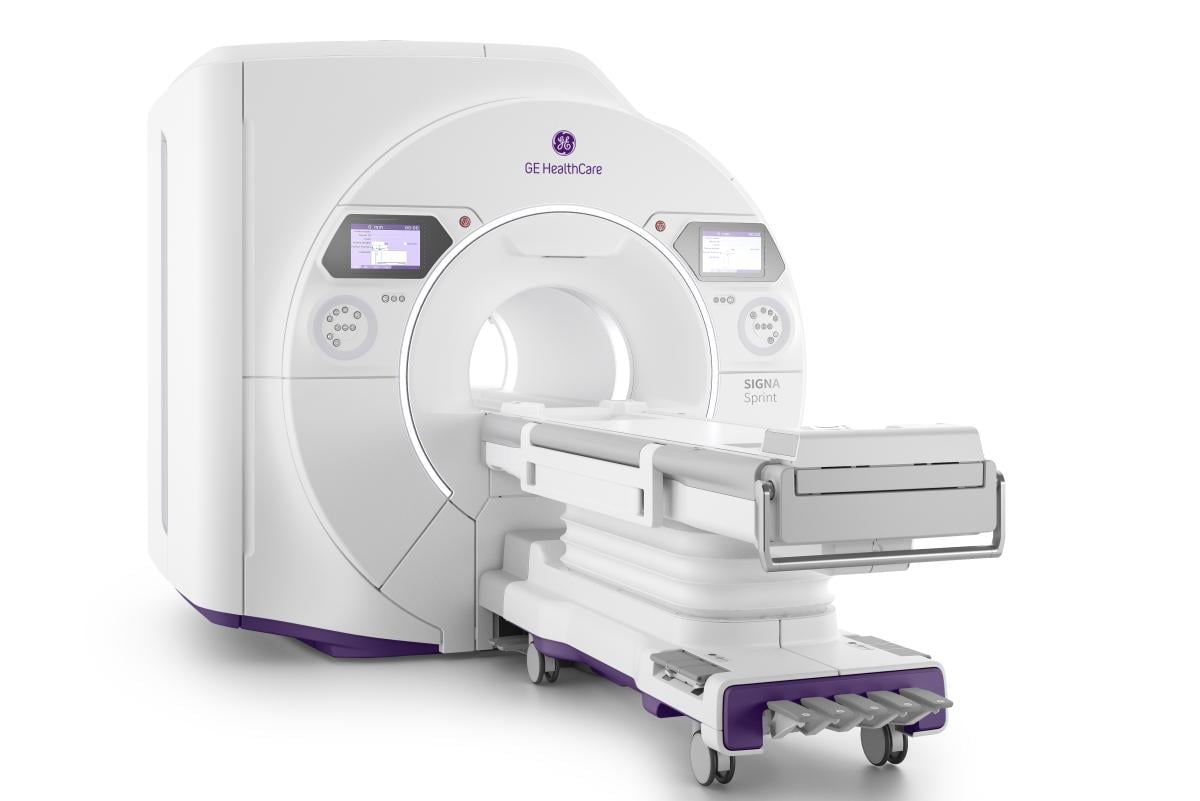
Signa Sprint, with a high-gradient performance of 65/200 per axis for fast imaging and exceptional image quality, is designed to deliver crystal-clear visualization of sub-millimetric structures and deep learning solutions to support diagnostics and treatment response monitoring in oncology patients. This new system supports clinicians’ shift to quantitative MRI and a deeper understanding of tissue characteristics, going beyond basic assessment of anatomy. The adoption of deep-learning reconstruction techniques for accelerated cardiac MRI aims to reduce the time and expertise needed to interpret scans and drive consistency and reliability.
“We are driven to push the boundaries of what’s possible in MRI with our ultra-premium segment, as our goal is to set a new standard in diagnostic research and precision care that allows for earlier clinical detection and treatment response,” said Kelly Londy, CEO, MR, GE HealthCare. “We are working to enhance diagnostic capabilities to provide highly accurate imaging with peace of mind for patients. We hope to help clinicians unlock new horizons for research in advanced imaging.”
Signa Sprint also is designed to optimize patient comfort, providing a 70 cm wide bore space, free-breathing capabilities, and blanket-like AIR Coils to improve the patient experience. This next-generation, high-performance platform is designed to benefit from built-in AI technologies – AIR Recon DL, Sonic DL and AIR x – which enable high-quality imaging even in the most demanding anatomies. Signa Sprint aims to support clinician goals of providing precision care through the entire care pathway, from the early diagnostic stage to treatment monitoring.
Learn more about SIGNA Sprint and other GE HealthCare MRI technologies at gehealthcare.com.
[1] Signa Sprint is 510(k) pending at US FDA. Not yet CE marked. Not available for sale in any region.
[2] World Health Organization (2025). CVDs statistics and treatment. World Health Organization. Retrieved from https://www.who.int/news-room/fact-sheets/detail/cardiovasculardiseases
[3] World Health Organization. (2025). Cancer statistics and treatment. World Health Organization. Retrieved from https://www.who.int/news-room/fact-sheets/detail/cancer


 May 01, 2026
May 01, 2026 









