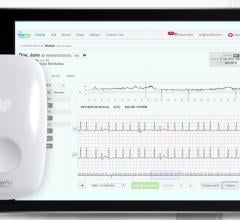
August 28, 2013 — AliveCor Inc. released a new AliveECG App for its Heart Monitor that features an Enhanced Filter, a noise cancelling technology that improves the visual output of the ECG. The AliveCor Heart Monitor is a mobile device-based ECG monitor for the iPhone 4, 4S and 5 that is used to record, display, store and transfer single-channel electrocardiogram (ECG) rhythms.
The Enhanced Filter aids in the reduction of noise commonly caused by the environment and/or by muscular vibrations that can result in non-readable or difficult to read rhythm strips. This major advancement improves visual quality and enables efficient interpretation. Ability to clearly delineate between normal sinus rhythm and arrhythmia is of utmost importance to both the physician and patient for proper treatment and management.
“The new technology makes it easier for patients using the device to record and send cleaner ECGs. This makes it easier and more efficient for me to diagnose the problem,” said Jeffery Olgin, chief of cardiology, University of California, San Francisco.
The new AliveECG App also features an improved menu that allows for easy navigation. Patients are able to annotate each ECG recording quickly by tapping on the current symptoms and/or activities. Tracking activities and symptoms provides greater context in which to interpret the ECG recording and can aid in the identification of arrhythmia triggers.
“AliveCor’s vision is to provide medical professionals and patients with cost effective quality solutions that deliver superior clinical utility,” said Euan Thomson, AliveCor CEO and president. “We have a robust product pipeline and plan to introduce new features, products, and services on a regular basis to further our vision.”
AliveCor’s Heart Monitor snaps onto the back of an iPhone to record, display, store and transfer single-channel ECG rhythms wirelessly with the corresponding, free AliveECG app. The Heart Monitor is U.S. Food and Drug administration (FDA) cleared and CE marked and makes ECG data accessible anytime, anywhere.
For more information: ww.alivecor.com


 July 22, 2025
July 22, 2025