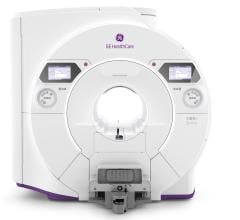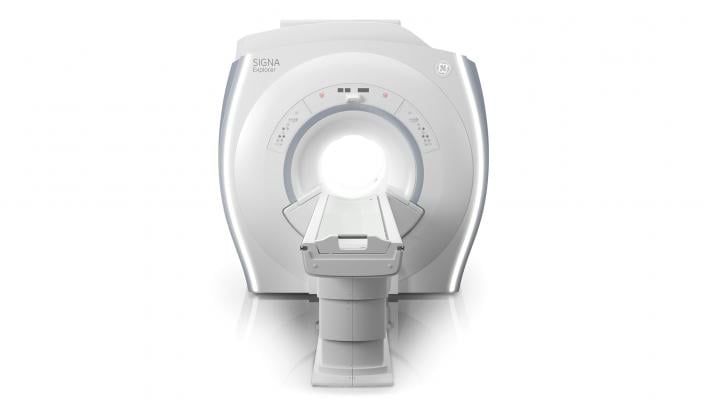
June 3, 2015 - GE Healthcare introduced a new in-field upgrade program for 1.5T magnetic resonance imaging (MRI) systems in most regions globally. A currently installed GE Healthcare 1.5T LCC magnet may be upgraded to the new Signa Explorer Lift so customers can benefit from the modernized patient comfort, workflow efficiency and diagnostic quality of this system. By upgrading, customers can potentially benefit from cost savings in multiple ways: up to 50 percent savings in construction cost, up to 50 percent savings in equipment cost in comparison to a new 1.5T system, and up to 30 percent increase in procedures due to potential increased throughput and referrals.
The Signa Explorer represents the latest offering from GE Healthcare, designed to help clinicians improve workflow, lower cost of ownership and improve patient comfort. This system was designed to use 34 percent less power than previous generation MRI systems and require a smaller footprint for installation. The upgrade to Signa Explorer covers a range of productivity-enhancing applications and new features to enhance patient comfort compared to previous generation systems - for example, SilentScan quiet technology. Signa Explorer and the upgrade also feature MAVRIC SL, which brings the power of MRI to patients with MRI-conditional metal implants by enabling visualization of soft tissue and bone near the implant.
"Orthopedic and Sports Medicine Center (OSMC) is very excited about our decision to purchase the Explorer upgrade for our existing magnet," said Donald Hammond, CEO of OSMC in Elkhart, Indiana, the first site to upgrade their system to the Signa Explorer Lift. "It is gratifying to know that a decision we made to choose GE over 10 years ago continues to give our organization significant benefits; including lower cost for upgraded versus new equipment as well as no construction cost associated with a new magnet while obtaining the most up-to-date clinical technology that provides our patients with exceptional quality and services available."
The upgrade will start to be made commercially available to the GE Healthcare MRI installed base during the coming months, in the United States and other countries where the Signa Explorer is available for sale (and has been approved, cleared or registered by the appropriate regulatory authorities). Offer may not be obtainable in all regions.
Upfront cost includes equipment and construction required for the equipment install and potential mobile cost during downtime. Actual costs will vary depending on your site's specific circumstances.
With the SIGNA Explorer Lift, the system may be able to scan 2-3 more patients per day due to new capabilities and productivity.
For more information: www.gehealthcare.com


 May 14, 2026
May 14, 2026