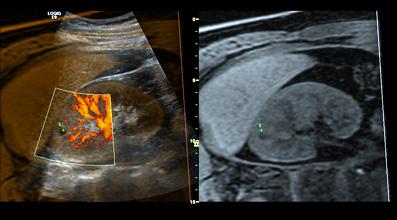
September 5, 2008 - GE Healthcare today said it launched a new ultrasound system for radiology and vascular applications, the LOGIQ E9, which fuses ultrasound images with images from other imaging modalities like CT and MR.
The LOGIQ E9 includes volume navigation, a tool incorporating two key components to maximize the system’s ultrasound architecture: fusion to combine the advantages of real-time ultrasound imaging with the high spatial and contrast resolution of CT, MR or PET; and a GPS-like technology to track and mark a patient’s anatomy during the ultrasound exam.
GE said the LOGIQ E9 helps address the biggest challenge in ultrasound radiology and vascular care – how to leverage clinical images from previous diagnostic imaging studies for interventional or diagnostic ultrasound procedures.
The LOGIQ E9 offers Scan Assistant, which aids clinicians to put the emphasis on diagnosis rather than on keystrokes. This tool allows a clinician to pre-program the actions most often performed, and then let the system do the detailed manipulations as the patient is scanned. The company said it improves exam time up to 54 percent, automatically inserts comments, completes measurements, steers color doppler, sets up imaging controls and modes, and improves ergonomics.
For more information: www.gehealthcare.com


 May 12, 2026
May 12, 2026 









