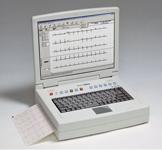
November 12, 2009 – In addition to administering patient tests and managing patient information, Welch Allyn’s new CP 300 offers a full-featured ECG with the convenience of a laptop, single-key operation, a full-color 15-inch flip-screen, flexible report formatting and a full-featured stress option.
The CP 300 is a noninterpretive 12-lead resting ECG with an optional interpretation software package that offers physicians a “second opinion.” It has a full alphanumeric keypad with dedicated function keys for quick access to common functions for easy patient data entry and waveform monitoring. In addition to PC-based hardware that runs on a Windows XP platform, the unit is equipped with an integrated high-resolution thermal printer that generates standard full-sized reports in customizable formats and an integrated graphical electrode test with checkpoints to aid electrode placement and check lead quality. The device also offers 40 GB of storage capacity to manage large volumes of test records.
The company said the CP 300 also offers a full-featured stress option with a complete exercise ECG test system. In addition to full disclosure of all 12 leads, it features dedicated function keys to streamline the testing process. Users can choose from six predefined, commonly-used stress protocols, or use the protocol editor to create their own. The device’s ST measurement reference point can be modified at any time during or after the test to calculate ECG measurements at regular intervals and/or display ST measurements on average complex and tabular views.
The system was developed by OEM Schiller AG.
For more information: www.welchallyn.com, www.schiller.ch


 May 08, 2026
May 08, 2026 









