
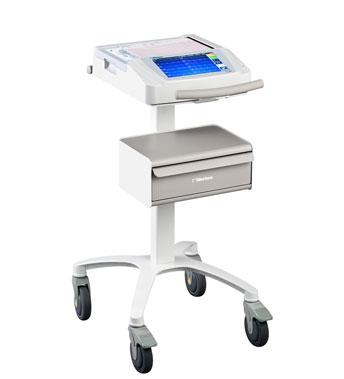
February 26, 2013 — Mortara Instrument Inc. began its worldwide release of its latest resting electrocardiograph (ECG), the ELI 280. The new system combines advanced ECG processing with an intuitive interface, enabling clinicians optimized efficiency in their acquisition of routine diagnostic ECGs.
Featuring a 10-inch tablet-style display with state-of-the-art touch technology, this system delivers good performance and ease-of-use to healthcare professionals in both clinics and acute care settings. The convenient onscreen keyboard features dedicated, colored keys for basic operations, making common tasks easier to perform and helping to streamline procedures.
As with other Mortara ECGs, the ELI 280 uses digital imaging and communications in medicine (DICOM) for standards-based data exchange with vendor-neutral clinical information systems. DICOM Modality Worklist (MWL) retrieves location-specific orders and schedules, simplifying workflow and speeding patient care. Acquired ECGs are then stored as discrete DICOM waveforms, fueling the clinical information systems’ advanced reporting solutions and automatic charge posting.
The ELI 280 features Mortara’s Veritas resting ECG interpretation algorithm. It also includes Mortara’s signature Best 10 feature and optional wireless ECG acquisition module (WAM).
For more information: www.mortara.com


 May 08, 2026
May 08, 2026 









