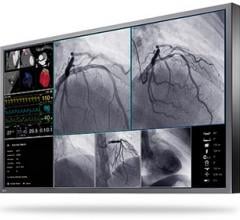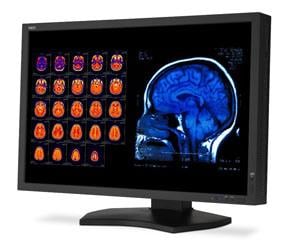
June 29, 2011 – The U.S. Food and Drug Administration (FDA) has given 510(k) market clearance of the 30-inch MultiSync MD301C4 medical diagnostic display for the displaying and viewing of digital images for diagnosis by trained physicians. The display is made by NEC Display Solutions of America.
The 4 megapixel (MP) MD301C4 was created for medical imaging and picture archiving and communication system (PACS) in hospitals, doctor offices, urgent care centers and other healthcare facilities. This medical-grade display includes patented technologies that make healthcare employees’ work more productive and accurate. It offers factory-adjusted DICOM calibration with self-monitoring brightness and, combined with the optional MDSVSENSOR2 color calibration sensor, the MD301C4 also supports standalone calibration and exact monitor matching, while enhancing calibration and white-point matching for outstanding DICOM GSDF conformance.
A partial list of features in the MultiSync MD301C4 follows:
- 30-inch (29.8-inch VIS) widescreen P-IPS panel
- 350 cd/m brightness (200 cd/m calibrated) with automatic luminance control that ensures consistent brightness for the life of the display
- Digital Uniformity Correction, which reduces screen uniformity errors and compensates for differences in color/grayscale and luminance across the screen
- 14-bit 3-D internal programmable look up tables (LUTs), which enable precise calibration with finely detailed, high-definition renderings of color images and crisper display of even the most delicate shadings and color differences
- AmbiBrightT ambient light sensor automatically adjusts the display’s brightness based on lighting conditions
- Two DisplayPort inputs, 2 DVI-D inputs and two up/three down USB hub with DisplaySync ProT, which can function as a keyboard, video and mouse (KVM) switch
- Picture-by-picture functionality
- Multi-directional controls, which allow for ease of use in either landscape or portrait modes
- XtraView+T for the widest viewing angles with minimal off-angle color shift
- Medical certifications, including CE-MDD 93/42/EC, UL 60601-1 and FDA 510(k)
- Restriction on Hazardous Substances (RoHS) compliant
For more information: www.necdisplay.com

 January 25, 2018
January 25, 2018