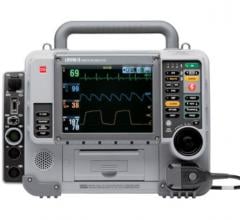
August 24, 2011 — Physio-Control Inc. announced commercial availability of the ReadyLink 12-lead electrocardiogram (ECG) device. It is a new U.S. Food and Drug Administration (FDA) 510(k)-cleared solution designed to improve care for heart attack patients and expand regional ST-segment elevated myocardial infarction (STEMI) systems.
ReadyLink enables basic life support (BLS) emergency medical service (EMS) providers to acquire and transmit a patient’s 12-lead ECG to hospitals using the Lifenet system. Lifenet is Physio-Control’s cloud-based data network for remote physician interpretation and decision support. This provides earlier insight into a chest pain patient’s condition, especially in rural areas with limited access to advanced life support (ALS) providers, and enables the most efficient routing for patient treatment.
The 2010 American Heart Association Guidelines for CPR and Emergency Cardiovascular Care emphasize the importance of a well-organized approach STEMI care to activate rapid access to reperfusion therapy.
STEMI is a type of heart attack that reduces or blocks circulation of blood to the heart; it can result in death or serious disability for the patient. ReadyLink expands a regional STEMI system’s reach by enabling BLS providers to rapidly identify STEMI patients with the assistance of trained clinicians; this starts the treatment clock earlier and reducing overall time to treatment.
ReadyLink requires a three-step process to capture and transmit a 12-lead ECG:
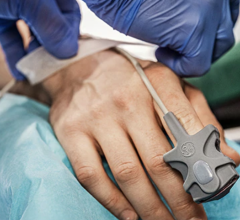
1) Prepare patient’s skin and place electrodes
2) One-button push to acquire 12-lead ECG and
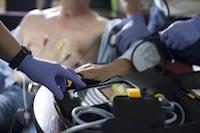
3) One-button push to transmit ECG to selected hospital facility via the Lifenet system.
Additional ReadyLink features include:
- All ECG leads visible on screen
- Full keyboard for easy patient data entry
- Backlit screen
- External AC/DC converter with rechargeable lithium ion battery
- Built-in modem
- Internal storage up to 60 ECGs
ReadyLink is being exhibited publicly for the first time at two pre-hospital conferences – Fire Rescue International (Atlanta, Aug. 26-27) and EMS World Expo (Las Vegas, Aug. 31-Sept. 2).
For more information: www.physio-control.com


 March 10, 2026
March 10, 2026