The U.S. Food and Drug Administration (FDA) today expanded the indicated use for the CoreValve…
Scottcare Corp.
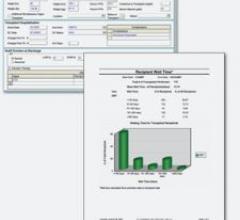
LUMEDX’s Cardiac Transplant solution is an integrated part of the company’s comprehensive suite…
Heavy duty, yet lightweight, the Detecto 6856 bariatric scale’s balanced design reportedly makes…
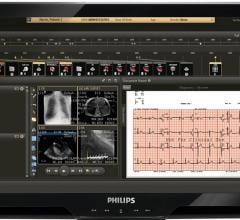
McKesson has released a new Horizon Cardiology product for the cath lab, the Holding Area…
Resurgent Health and Medical is introducing its new CleanTech IC hand washing system, which…
Medical gas and vacuum products maker Ohio Medical Corp.
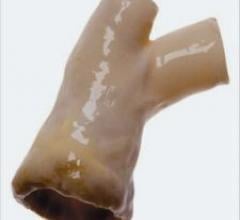
The FDA cleared CryoLife’s CryoValve SG pulmonary human heart valve processed with the company’s…
The R Series defibrillator for hospitals aims for simplicity and operational readiness to help…
Vascular Solutions' Jiffy Wire, an 80 cm 0.035-inch specialty guide wire, was launched in July…
St. Jude Medical said the FDA cleared its SJM Confirm implantable cardiac monitor (ICM), a…
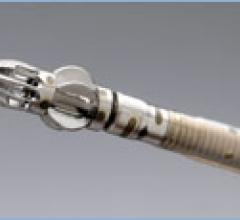
The ClariTEE miniaturized transesophageal echocardiography probe facilitates episodic monitoring…
March 23, 2009 - Pathway Medical Technologies Inc. today said the FDA granted the company 510(k…
The LATITUDE Patient Management System by Boston Scientific has FDA approval to be used in…
Calgary Scientific Inc. Medical Group (CSIMG) announced the receipt of FDA 510(k) clearance for…
Boston Scientific Corp.
The BariMaxx II Power Drive System is a 1,000-pound capacity expandable side exit bariatric bed…


 June 12, 2014
June 12, 2014