July 20, 2007 – Oraganogenesis Inc. announced that it has received regulatory approval in Canada and Europe for BioSTAR, a device that incorporates a bioresorbable collagen scaffold used to treat patent foramen ovale (PFO), a hole in the heart that is usually asymptomatic.
Source: Wall Street Journal Online July 20, 2007 - U.S. doctors implanted fewer coronary stents in June than any other ...
July 19, 2007 – Abbott announced that the first patient was enrolled in its XIENCE V SPIRIT WOMEN clinical trial, the ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
July 20, 2007 – Despite forecasts by analysts at Thomson Financial that Boston Scientific Corp. would earn $150 million ...
July 19, 2007 – AtCor Medical Holdings Ltd., the developer and marketer of the SphygmoCor system, which noninvasively ...
July 18, 2007 - Abiomed Inc. announced that its Impella 2.5 and Impella 5.0 circulatory support technologies, new ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
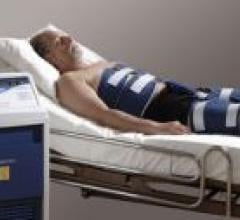
Cardium Therapeutics and its operating unit, InnerCool Therapies, have released its new CoolBlue surface temperature ...
July 18, 2007 - Cardium Therapeutics and its operating unit, InnerCool Therapies, have released its new CoolBlue surface ...
July 18, 2007 - An analysis based on computerized simulation models suggests that the lifetime risk of cancer associated ...
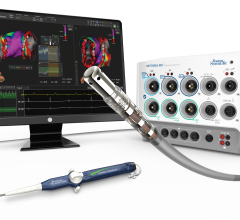
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
July 18, 2008 - Cardium Therapeutics announced that the FDA has given it clearance for fast track designation to its ...
July 18, 2007 - In a related study of over 25,000 initially healthy women, higher triglyceride levels measured after not fasting is associated with an increased risk for cardiovascular events, but his association was not found for triglyceride levels measured after fasting, according to a study in the July 18 issue of JAMA.
July 17, 2007 - Medtronic Inc. announced launch of Reveal XT, an Insertable Cardiac Monitor that features long-term and continuous monitoring of Atrial Fibrillation (AF), in the European market as it recently received CE (Conformité Européenne) Mark.
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
July 17, 2007 – LDS Hospital will become certified to implant the CardioWest temporary Total Artificial Heart (TAH-t) ...
July 17, 2007 - St. Jude Medical Inc. today announced that it received regulatory approval from the Japanese Ministry of Health, Labor and Welfare and reimbursement approval for its first CRT-D systems to be marketed in Japan.
July 17, 2007 - Heart Imaging Technologies (HeartIT) announced today that medical images can be viewed on Apple’s new iPhone.

 July 19, 2007
July 19, 2007