The landscape of cardiovascular care is changing. In the cath lab alone, the shift in volume and type of diagnostic and interventional procedures has broadened the lab’s scope, increased demand and raised expectations.
LUMEDX Corp. offers the CardioPACS 5.0, the latest version of its cardiology PACS software. The multimodality, Web ...
Cook Medical has received approval from the FDA for its Zenith TX2 Thoracic TAA Endovascular Graft, marking the expansion of Cook Medical’s portfolio of devices to treat aortic diseases.
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
St. Jude Medical Inc. recently released its EnSite System Version 8.0, software designed to intuitively visualize the ...
Nihon Kohden America Inc. said its Prefense Early Detection and Notification System received 510(k) clearance from the ...
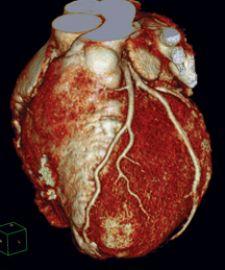
Angiography systems have been a diagnostic mainstay in cardiac cath labs for more than 50 years, playing a pivotal role in the diagnosis and treatment of heart and vascular diseases. Replacing imaging intensifiers with flat panel detectors (FPD) for angiography has broadened digital imaging’s role in the cath lab.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
September 3, 2008 - Endologix Inc. yesterday said six-year patient follow-up data from the company’s Powerlink System ...
The FDA recently cleared Boston Scientific Corp.’s PROMUS Everolimus-Eluting Coronary Stent System and Abbott Vascular’s ...
AngioScore Inc. has introduced longer and larger AngioSculpt PTA Scoring Balloon Catheters for the treatment of peripheral artery disease (PAD).
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
The R Series defibrillator for hospitals aims for simplicity and operational readiness to help improve in-hospital ...
While the primary reason hospitals adopt new technology is to increase patient care and produce better outcomes, there are many other factors that also affect purchasing decisions.
September 4, 2008 – St. Elizabeth, in Cincinnati, OH, which serves more than 300 stroke patients annually, is the first ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
September 3, 2008 - Edwards Lifesciences Corp. today said it received FDA approval for the Carpentier-Edwards PERIMOUNT ...
September 2, 2008 - The safety debate about drug-eluting stents (DES) is still ongoing and in these times of uncertainty ...
Edwards Lifesciences Corp. produces the Carpentier-Edwards The PERIMOUNT Magna mitral valve was launched in Europe in September 2005. It incorporates features of the Carpentier-Edwards PERIMOUNT mitral valve, which has demonstrated 16 years of durability, including the treatment of the bovine pericardial tissue leaflets with the Carpentier-Edwards ThermaFix process. September 2008

 September 03, 2008
September 03, 2008




