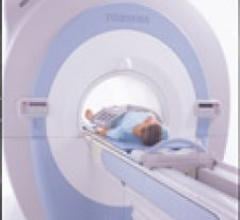
Toshiba's Vantage Titan MRI is designed to capture higher resolution heart images, while capturing deeper imaging of the anatomy. This is possible due to the center eight elements of the cardiac coils being smaller and the outer eight elements of the cardiac coils being larger on the Vantage Titan according to Toshiba.
February 11, 2008 - The American Society of Echocardiography (ASE) has issued a new consensus statement for interpreting ...
February 11, 2008 - Boston Scientific Corp. today announced that the first patient has been enrolled in its PROENCY ...
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
A third round of software enhancements has been made to the iLab System 1.3, an ultrasound imaging system designed to strengthen physician confidence in the diagnosis and intervention of percutaneous coronary intervention (PCI) and increase workflow efficiency.
February 11, 2008 - The first patient was successfully treated in AtriCure’s ABLATE clinical trial designed to evaluate ...
CryoLife Inc.'s CryoValve SG pulmonary human heart valve is processed with the company’s proprietary SynerGraft technology, providing a valve replacement option for children born with heart defects or for replacement surgery.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
February 8, 2008 – CryoLife Inc. said it received FDA 510(k) clearance for its CryoValve SG pulmonary human heart valve ...
February 8, 2008 - Emageon received first place in the “Best in KLAS” Top 20 Report for the Cardiology PACS nomination ...
February 8, 2008 - The FDA granted Volcano Corp. 510(k) clearance for its s5-Revo and s5-FFR (fractional flow reserve) options, allowing physicians access phased Array IVUS, Rotational IVUS and integrated FFR on the same integrated IVUS platform.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
February 7, 2008 - Volcano Corp. said the FDA cleared the s5-Revo and s5-FFR (fractional flow reserve) options, enabling ...
February 8, 2008 - CryoLife Inc. received 510(k) clearance from the FDA for its CryoValve SG pulmonary human heart valve processed with the company’s proprietary SynerGraft technology, providing a valve replacement option for children born with heart defects or for replacement surgery.
Emageon's HeartSuite offers cardiovascular information management, hemodynamic monitoring, advanced visualization and content management. Emageon’s architecture combines HeartSuite with radiology and other medical specialties designed to offer a comprehensive patient–centric view of the visual medical record. One suite of integrated, enterprise cardiology tools includes:
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
February 8, 2008 - The FDA granted Volcano Corp. 510(k) clearance for its s5-Revo and s5-FFR (fractional flow reserve) options, allowing physicians access phased Array IVUS, Rotational IVUS and integrated FFR on the same integrated IVUS platform.
McKesson’s ECG Management module, a component of Horizon Cardiology, is a fully integrated, Web-enabled ...
February 7, 2008 - Abiomed Inc. reported its third quarter fiscal 2008 revenue reached $16.0 million, up 24 percent ...

 February 10, 2008
February 10, 2008