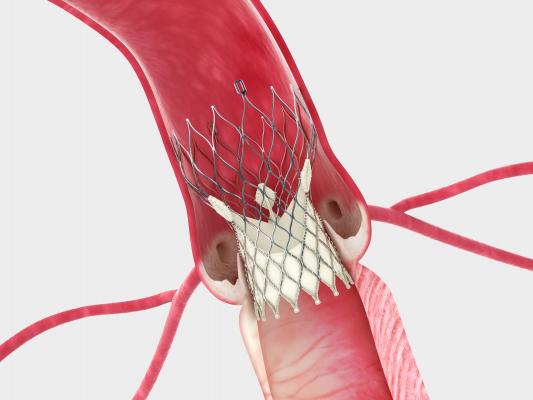
May 20, 2014 — In a surprise move, Edwards Lifesciences Corp. and Medtronic reached an agreement this week to settle all outstanding patent litigation between the companies, including cases related to transcatheter heart valves. The agreement will result in the dismissal of all pending cases or appeals in courts and patent offices worldwide, and includes a provision that the parties will not litigate patent disputes with each other in the field of transcatheter valves for the eight-year duration of the agreement.
The companies have been embroiled in patent litigation over their transcatheter aortic valve replacement (TAVR) systems in Europe and the United States for the past several years. There has been concern by many top physicians that the competition would hinder patient access to these life-saving therapies. This concern was heightened with recent clinical trial data showing Medtronic’s CoreValve out-performed surgical valve replacement in some patients. However, this was tempered when Edwards filed an injunction against the sale of the valve in the U.S. market.
Under the agreement, Medtronic will pay Edwards a one-time payment of $750 million, and ongoing royalty payments through April 2022 based on a percentage of CoreValve sales, in payments of no less than $40 million annually.
In addition to settling the pending lawsuits with cross-licenses, Medtronic and Edwards have agreed that neither party will sue the other for patent matters anywhere in the world for eight years in the field of aortic and all other transcatheter heart valves.
"This agreement brings to an end years of disputes between our companies related to TAVI [transcatheter aortic valve implantation] patents, and allows both companies to make their respective therapies available to physicians and patients around the world," said John Liddicoat M.D., president of the structural heart business at Medtronic. "With this resolution, we are pleased that Medtronic will be able to continue to provide the CoreValve system, as well as other products, to patients who need them in the U.S. and abroad without the overhang of any potential injunction or additional damages."
Edwards will contribute $50 million from the settlement to the Edwards Lifesciences Foundation to support efforts to improve patient care, raise disease awareness and educate clinicians, such as through its Every Heartbeat Matters philanthropic program.
"We are pleased to reach an agreement that preserves physician choice while also recognizing Edwards' leadership in pioneering the transcatheter heart valves that are chosen most often by physicians worldwide. This agreement allows us to move forward, fully dedicating our time and resources to helping patients," said Michael A. Mussallem, Edwards' chairman and CEO.
For more information: www.edwards.com, www.medtronic.com


 April 24, 2026
April 24, 2026 









