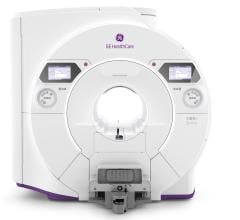
May 20, 2014 — GE Healthcare and Tesla Engineering Ltd. are collaborating to produce 7T human whole-body magnetic resonance imaging (MRI) scanners. The companies made the announcement at the joint meeting of the International Society for Magnetic Resonance in Medicine (ISMRM) and the European Society for Magnetic Resonance in Medicine and Biology (ESMRMB) in Milan.
Ultra high-field MRI systems are used for scientific and medical research, primarily for morphological and functional imaging of the brain while steadily expanding to all anatomies. Researchers using ultra high-field scanners hope their work will contribute to earlier disease detection, more accurate diagnosis and increased effectiveness of investigational therapies for disorders and diseases such as cancer, stroke, epilepsy, drug abuse, Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, attention deficit disorder, multiple sclerosis, head injury, visual disorders and schizophrenia.
Tesla Engineering Ltd. is currently building a new factory in Storrington, U.K., to manufacture the 7 Tesla magnets. They have recruited several key engineering and management staff with extensive experience in the design and manufacturing of ultra high-field magnets. The first unit from the new production line is committed to GE and scheduled to ship in late 2015.
“This agreement will enable GE to deepen and broaden our collaborations with leading MRI academics and visionaries, sharing our collective expertise and missions in technology, science and medicine,” said Richard Hausmann, president and CEO of GE Healthcare, MR. “The GE 7T community in the U.S., Europe and Asia has already demonstrated breakthrough observations and understanding of Alzheimer’s disease, traumatic brain injury and cognitive physiology. Together, we will continue to push the frontiers of MRI for neuroscience and other applications.”
“Tesla is very pleased to collaborate with GE and to take a leadership position in the continuing evolution of MR magnet technology,” said Mike Begg, group managing director, Tesla Engineering Ltd. “This is a solid business opportunity for us that complements and builds upon our existing strengths in superconducting magnet technology, as well as our strong core technology positions in cryogenics.”
For more information: www.gehealthcare.com, www.tesla.co.uk


 May 14, 2026
May 14, 2026