The annual Transcatheter Cardiovascular Therapeutics (TCT) meeting brings many cutting-edge technologies to the forefront during its five days of clinical sessions. This is my editor’s choice of the most innovative technologies discussed during TCT 2012.
Holograms to Guide Interventions
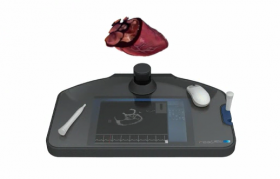
Navigation in the cath cab has been greatly enhanced with 3-D anatomical images projected on overhead screens or used as fused overlays on live angiography. However, the images remain displayed on 2-D screens and there is still reconstruction of the images in true 3-D in the physician’s head. New technology developed by an Israeli company called RealView now allows true 3-D holograms to be projected in mid-air above patients in the cath lab.
The RealView Holographic Imaging System can project 3-D images from datasets created by computed tomography (CT), magnetic resonance imaging (MRI), rotational angiography or 3-D ultrasound such as transesophageal echo (TEE). The mobile console has a table platform that can be positioned over a patient in the cath lab so the interventionalists have a close-up view of the anatomy and can interact with it. Using mid-air hand motions, the hologram can be rotated in any direction, and using a hand-held slicing device, images can be cut through to reveal specific anatomical views.
The technology is expected to have a major impact on more complex interventional procedures including structural heart and electrophysiology (EP) mapping and ablation navigation. Procedures such as left atrial appendage (LAA) occlusion and mitral valve repair are extremely difficult or impossible to do without 3-D imaging, and this technology is expected to greatly enhance current imaging options.
“3-D holography in the cath lab has several objectives,” said Elchanan Bruckheimer, MBBS, director, pediatric cardiac catheterization, Schneider Children’s Medical Center, Petach Tikva, Israel, who helped develop the technology. “With this you don’t have to take three or four images and reconstruct it in your head. We use these images to navigate, but it is very different to navigate in the heart using these holograms.”
He said the system will be tested in feasibility studies in 2013.
MRI Guidance in the Cath Lab
As interventional procedures become more complex, the amount of angiographic X-ray time and radiation dose received by the patient is rapidly rising. However, MRI may be able to offer a replacement imaging modality for fluoroscopy. It would not only eliminate radiation, but also greatly improve anatomical imaging without the use of radiation or contrast. MRI transcatheter interventions are already being performed by the National Institutes of Health (NIH), which is now collaborating with Children’s Memorial Hospital in Washington, D.C., to build an MRI-based cath lab in 2013.
“MRI has a lot more image data than angiography,” said senior investigator Robert Lederman, M.D., cardiovascular intervention, cardiovascular and pulmonary branch, NIH, in Bethesda, Md. “It’s pretty compelling for pediatrics, and it allows you to visualize tissue damage while you are working.”
For example, he said, chronic total occlusions (CTOs) are difficult to treat because angiography only shows contrast in the portion of the vessel that is not blocked. Pushing wires or other devices into a CTO is done somewhat blindly, since an operator cannot know with certainty where the wire is going, even with a CT or MRI reference image. But with live MRI imaging, he said, all the soft tissue anatomy is visible, so even the occluded sections of vessels are seen. Catheters and wires appear as glowing green objects in the live MRI images, so they are more easily seen than on angiography.
Lederman said the technology also has tremendous application in pediatric cardiology, where radiation is a major concern. Applications for pediatric cardiology include closure of ventricular septal defects (VSDs), shunts and better visualization for transapical delivery of therapies and closure devices.
An advantage of MRI is that the contrast levels can be easily altered easily to better visualize certain types of anatomy, instead of just seeing an X-ray artio-lumingram. Lederman said some alterations had to be made to make standard devices easier to visualize under MRI. Balloons, for example, have to be modified and are filled with gadolinium contrast agents so they can be visualized, since air-filled balloons are very difficult to see.
The ability to immediately visualize tissue damage under MRI may also hold promise for improved EP ablation procedures, where the lesions can be seen as they are created.
NIH and Siemens Medical Systems have a collaborative research and development agreement for interventional cardiovascular MRI.
Preventing Diastolic Fluid Buildup in Heart Failure
At the TCT sessions, a new structural heart interventional device was introduced that may help reduce the symptoms of diastolic heart failure. DC Devices is in preclinical testing of the Intra-Atrial Shunt Device (IASD), which creates a left-to-right atrial shunt to relieve left atrial pressure. The operator gains access to the right atrium from the inferior vena cava, makes a transeptal puncture, balloons the hole and then implants the device. The device is a permanent implant that uses a self-expanding nitinol framework to ensure the hole does not close.
The theory is that the heart stiffens and reduces its elasticity due to pressure buildup. The device is supposed to relieve the pressure and prevent fluid backup in the lungs. If cleared, it will be the first device to address diastolic heart failure, which is currently only managed with pharmacological agents.
The device is currently being used in a small pilot trial in Europe to show proof of concept. It is expected to enter human trials in Europe in 2013, in hopes of gaining CE mark clearance. “We are not curing diastolic heart failure — no one can — but we are correcting one of the main symptoms of diastolic heart failure,” said George Fazio, president and CEO of DC Devices.
Sympathetic Nerve Ablation to Treat Heart Failure
The original developers of renal denervation therapy to short circuit the sympathetic nervous system to reduce hypertension have expanded their concept to other disease states. They sold their previous startup company to Medtronic, and their new startup company, Cibiem, is working on a similar radiofrequency (RF) ablation catheter for use in the carotid artery to kill the sympathetic nerves that play a role in heart failure.
The Carotid Body Modulation (CBM) system is a minimally invasive, catheter-based treatment designed to treat sympathetic nervous system-mediated diseases such as hypertension, heart failure, diabetes and renal failure. The therapy focuses on the modulation of the carotid body, which is a key chemosensory located at the fork of the carotid artery that helps regulate respiratory activity.
Cibiem recently announced a $10 million Series A round of financing to support the development of CBM.
Renal Denervation
The idea of using a balloon catheter equipped with electrodes to kill the sympathetic nervous system controlling renal artery vasoconstriction to help cure pharmacologically uncontrolled hypertension has moved from a science project discussion into the mainstream. This was abundantly evident at TCT 2012, where there were numerous presentations on Medtronic’s Symplicity catheter and its ongoing U.S. Food and Drug Administration (FDA) pivotal trial.
Several new startup companies and major vendors announced they too were developing renal denervation therapy devices. New entrants to this device development arena include Covidien, Boston Scientific, St. Jude Medical, CardioSonic, ReCor Medical and Kona Medical. Boston Scientific recently paid about $425 million to acquire Vessix Vascular and its V2 denervation system, and showed it as a work-in-progress for the first time at TCT.
The new systems being offered by newcomers offer potential improvements over the Symplicity system, which requires the balloon to be moved and rotated to perform several ablations. For example, the Covidien OneShot system uses a single, spiral electrode so the entire renal artery can be ablated at once. On either side of the electrode are small irrigation ports to cool the tissue as the lesion is formed, similar to EP-irrigated ablation catheters. The balloon can also be filled with contrast to show if it is fully opposed before beginning the ablation.
Robotics in the Cath Lab
Several TCT sessions emphasized the impact of the da Vinci robotic system on surgery and compared it to the same level that image robotics will have in the cath lab. In the past year two cath lab robotic systems were cleared by the FDA: the Corindus CorPath system for coronary procedures and the Hansen Magellan system for peripheral interventions. Both received a good amount of attention in sessions and on the show floor.
Sharing his experience with using the Corindus system, Giora Weisz, M.D., director, clinical cardiovascular research, Columbia University Medical Center, New York, said the system allows operators to sit in a chair in a lead-lined booth, so they do not have to lean over a patient while wearing lead aprons, or expose themselves to radiation from the imaging system. Also, the system’s joystick controllers allow very precise, controlled millimeter increment movements that are not possible by hand, regardless of an operator’s experience.
“It does not have a tactile feel, but we can’t do procedures with our eyes closed by just using tactile feel, but we can learn to do procedures with just our eyes,” Weisz said. “We are doing more and more complex cases each day.”
He said there is a learning curve involved with the robotic systems, but the accuracy in catheter manipulation it allows is unprecedented. He reports using the system for the majority of his cases now.
Noninvasive CT-FFR
With clinical trial data to back the technology (the DeFACTO trial results were released last August), there were numerous mentions of HeartFlow’s computed tomography fractional flow reserve (CT-FFR) software to assess the hemodynamic impact of coronary stenoses. Currently, catheter-based FFR measurements can be done in the cath lab, but take time and require the use of adenosine injections. The CT-based software uses supercomputing fluid dynamic algorithms to measure FFR for the entire coronary tree. The software also allows placement of virtual stents to assess their impact on coronary flow both at the lesion and downstream.
The DeFACTO trial’s principal investigator James K. Min, director of cardiac imaging research and co-director of cardiac imaging at the department of medicine, imaging and biomedical sciences at Cedars-Sinai Heart Institute in Los Angeles, presented at TCT. He said the technology has the potential to eliminate the current diagnostic testing chain of ECG followed by a nuclear scan or stress echo, and then a diagnostic catheterization. He said CT-FFR could be a paradigm shift in how coronary lesions are assessed noninvasively, and stenting procedures could be preplanned before the patient enters the cath lab. Min said this could go a long way to help lower healthcare costs by eliminating several diagnostics tests. CT-FFR can also be used to assess which patients might benefit from pharmacological treatment alone, eliminating diagnostic catheterizations and the interventionalists’ temptation to stent what appear to be angiographically significant lesions, but may not have any restriction on flow.
“The current paradigms do not really identify obstructive coronary artery disease. The National Cardiovascular Data Registry (NCDR) identified high numbers of catheterizations that had no serious disease,” Min explained. “The current paradigm is not ideal and results in unnecessary catheterizations.”
Martin Leon, M.D., director, center for interventional vascular therapy, Columbia University Medical Center / New York-Presbyterian Hospital, and founder of TCT, predicted in one of his presentations that CT-FFR will a become standard diagnostic test in the future. He explained it has the potential to decrease costs by eliminating the need for numerous tests and may eliminate the need for diagnostic catheterizations.


 May 14, 2026
May 14, 2026