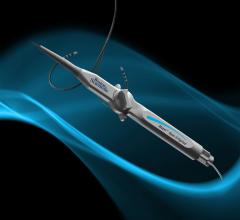
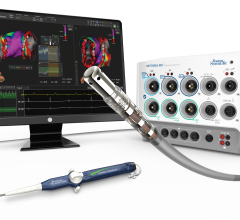
February 25, 2013 — The U.S. Food and Drug Administration (FDA) granted final approval for the Biotronik Lumax 740 DX ...
Atrial Fibrillation
This channel includes news and new technology innovations for the treatment of atrial fibrillation, also referred to as AF or afib. AF is a cardiac arrhythmia caused by irregular and often rapid heart rate. It is caused by the upper chambers (the atria) beating irregularly and uncoordinated with the lower ventricle chambers of of the heart. Symptoms include weakness with heart palpitations and shortness of breath. The conditional can lead to an increased risk of stroke and heart failure. AF episodes can cause the blood in the atria to stagnate and form clots, usually within the left atrial appendage (LAA). The clots can flow to the brain and cause a stroke. Treatments include anticoagulation therapy to dissolve clots, catheter or surgical ablation and LAA occlusion.
To help identify trends and find out what DAIC readers are interested in, the magazine takes note of what they click on ...
January 21, 2013 — The first patient has been treated in the Boston Scientific Corporation ZERO AF clinical trial to ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
January 21, 2013 — At the Boston Atrial Fibrillation (AF) Symposium 2013, Philips Healthcare introduced its latest ...
January 18, 2013 — nContact Inc. announced that the U.S. Food and Drug Administration (FDA) has cleared the Company’s ...
January 8, 2013 — The results of a recent study presented yesterday at the American Heart Association Scientific ...
For over a decade, the cardiac cryoablation industry has seen little in the way of technological advancements. Yet ...
November 15, 2012 — The U.S. Patent and Trademark Office has awarded Biomedical Systems U.S. Patent Number 8301236 for ...
November 9, 2012 — Bristol-Myers Squibb Co. and Pfizer Inc. announced that the reductions in stroke or systemic embolism ...
October 8, 2012 — The U.S. Food and Drug Administration (FDA) has granted Clinical Laboratory Improvement Amendments ...
In the United States, the options currently available for cardiac ablation use thermal mechanisms to ablate tissue and ...
October 2, 2012 — A novel blood thinner recently approved by the U.S. Food and Drug Administration (FDA), Pradaxa ...
October 1, 2012 — Bristol-Myers Squibb Company and Pfizer Inc. announced that the U.S. Food and Drug Administration (FDA ...
September 19, 2012 — This past week, surgeons and cardiologists at Sentara Heart Hospital were the first in the world to ...
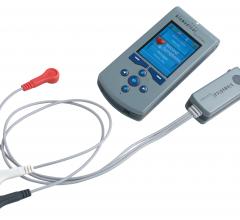
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
September 13, 2012 — Janssen Research & Development LLC announced that it has submitted the complete response to the U.S ...

September 11, 2012 — Since the previous consensus document was published in 2007, catheter and surgical ablation of atri ...
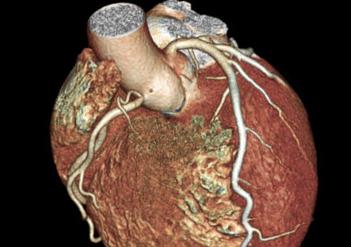
September 10, 2012 — Atrial fibrillation (AF) is a common complication of cardiac surgery, occurring in 10 to 40 percent ...


 February 25, 2013
February 25, 2013