February 14, 2024 — Efemoral Medical, developer of advanced interventional bioresorbable therapies, today announced that the United States Food & Drug Administration (FDA) has granted its novel Efemoral Vascular Scaffold System (EVSS) Breakthrough Device status for the treatment of de novo or restenotic lesions of the infrapopliteal arteries in patients with Chronic Limb Threatening Ischemia (CLTI).
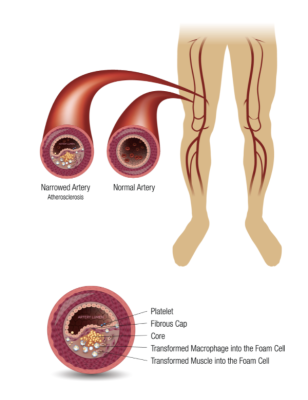
The Efemoral Vascular Scaffold System (EVSS) offers a new approach to treating peripheral arterial occlusive disease (PAOD) by addressing the specific anatomic challenges and complex biomechanics of patients with athero-occlusive disease in the leg. Using multiple, serial, intravascular scaffolds, the patented FlexStep Technology combines flexibility with support to open clogged vessels and sustain healthy blood flow while accommodating tortuosity and skeletal movement. Formulated with sirolimus antiproliferative drug elution, the bioresorbable scaffolds restore normal vessel diameter at the time of the procedure, deliver therapeutic benefits across all lesion lengths and morphologies, and maintain durable patency while leaving no permanent implant behind. The device designed for above-the-knee (femoropopliteal) intervention is currently being tested in a first-in-human trial, EFEMORAL I, in investigative sites in New Zealand and Australia. Encouraged by the early clinical results, Efemoral Medical is now developing an additional device for treating below-the-knee (infrapopliteal) arteries in patients with CLTI.
"Diseased human arteries are most simply, reliably, and successfully treated with drug-eluting, balloon-expandable stents," said Lewis B. Schwartz, MD, Co-Founder and CMO of Efemoral Medical, "however, it is extremely challenging to implant these permanent devices in the long and twisting arteries of the lower extremities. The EVSS uses a unique design of alternating, dissolvable, drug-eluting scaffolds that, for the first time, allows the long arteries of the legs to be treated with the same, effective, drug-eluting technology proven to be successful in multiple other vascular beds."
The Breakthrough Devices Program is reserved for novel medical technologies that potentially provide for more effective diagnosis and/or treatment of life-threatening or irreversibly debilitating diseases. It is intended for patients to enjoy more timely access to novel devices by expediting their development, assessment, and review.
Vascular disease of the lower extremities, known as "peripheral arterial occlusive disease" (PAOD), is a virtual epidemic, afflicting approximately 10% of the global population over the age of 50 and 20% of the population over the age of 70.1 Symptomatic PAOD causes poor physical health, loss of mobility and independence, decreased quality of life, premature functional decline and early mortality.2 An estimated 10% of patients with PAOD have its most severe form: chronic limb-threatening ischemia (CLTI). CLTI arises when the lesions of PAOD have become so numerous and severe that the blood flow to the foot is inadequate to sustain its viability. Only about half of all diagnosed patients will be alive after six months, and only about half of the survivors will have intact limbs. Given this dismal outcome, the prognosis of CLTI is worse than most cancers.3,4
"PAOD has a significant negative impact on the health of as many as 20 million Americans," said Christopher Haig, Co-Founder and CEO of Efemoral Medical.5 "It is especially devastating for those 10% of patients suffering from its most severe form, CLTI. We are extremely gratified that the FDA has recognized the potential of the EVSS to benefit these patients by granting Breakthrough Device status to our device."
For more information: www.efemoralmedical.com
References:
1 Eid MA, Mehta K, Barnes JA, Wanken Z, Columbo JA, Stone DH, et al. The global burden of peripheral artery disease. J Vasc Surg. 2023;77:1119-26.
2 McDermott MM, Liu K, Ferrucci L, Tian L, Guralnik JM, Liao Y, et al. Decline in functional performance predicts later increased mobility loss and mortality in peripheral arterial disease. J Am Coll Card. 2011;57:962-70.
3 Engelhardt M, Boos J, Bruijnen H, Wohlgemuth W, Willy C, Tannheimer M, et al. Critical limb ischaemia: Initial treatment and predictors of amputation-free survival. Eur J Vasc Endovasc Surg. 2012;43:55-61.
4 Mustapha JA, Katzen BT, Neville RF, Lookstein RA, Zeller T, Miller LE, et al. Determinants of long-term outcomes and costs in the management of critical limb ischemia: A population-based cohort study. J Am Heart Assoc. 2018. DOI: 10.1161/JAHA.118.009724.
5 Eid MA, Mehta K, Barnes JA, Wanken Z, Columbo JA, Stone DH, et al. The global burden of peripheral artery disease. J Vasc Surg. 2023;77:1119-26


 May 13, 2026
May 13, 2026 









