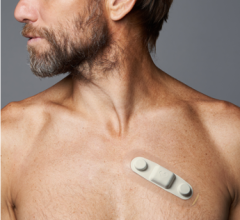
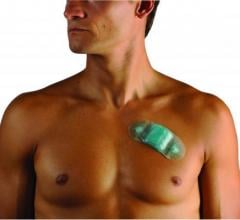
Biotres device
April 12, 2022 – Biotricity, a medical diagnostic and consumer healthcare technology company, will officially launch the commercial sales of its FDA cleared, wireless wearable cardiac monitoring device, Biotres, in April 2022. The product has been available for pre-orders to physicians, medical offices, hospitals and individual use since late-February 2022.
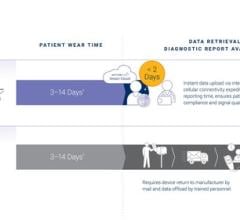
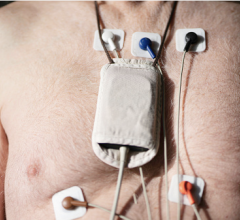
Biotres is the company’s revolutionary technology that represents the future of Remote Patient Monitoring and the delivery of real-time diagnostic data. It serves as a three-lead device designed to continuously record electrocardiogram (ECG) data for early detection of cardiac arrhythmias, disrupting the conventional one-lead patch Holter monitor which takes longer analysis time and diagnosis time.
Biotres received FDA 510(k) clearance in January 2022, opening up a new market and expanding Biotricity’s total addressable market to $5.7 Billion. Since taking pre-orders last month, the Company has experienced strong demand for Biotres from existing customers. The Company has plans to roll out additional complementary products throughout 2022, following the same approach as Biotres. Each solution will grow the company’s TAM, creating new revenue streams for existing customers while expanding its customer base.
Dr. Waqaas Al-Siddiq, Biotricity Founder and CEO, commented, “After months of anticipation, our Biotres product is finally available for sale. Judging by the amount of pre-order interest we’ve seen since February, Biotres can provide millions of individuals with an early detection method to easily and efficiently record cardiac arrhythmias. We are optimally positioned to penetrate a $2 billion-dollar holter market that needs a faster and more efficient approach, critical to improving patient lives.”
For more information: https://www.biotricity.com/biotres/


 March 31, 2025
March 31, 2025