Echopixel's true 3-D rendering of a ASD with a virtually implanted Amplatzer occluder to check sizing on a 3D echo prior to a procedure.
I saw several elements of what will likely make up the next generation of echocardiography imaging systems at the the American Society of Echocardiography (ASE) 2017 scientific sessions in June. I have seen or heard of these technologies for the past few years, but for the first time I saw them all moving forward and starting to coalesce into real, tangible products.
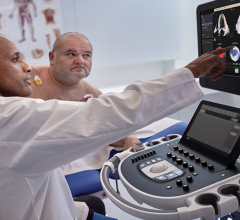
• Real 3-D Image Navigation in the Cath Lab: One of the coolest new imaging technologies I have seen in recent years is true 3-D imaging offered by EchoPixel. The technology allows 3-D image datasets to be displayed in actual 3-D from computed tomography (CT), magnetic resonance imaging (MRI) and 3-D ultrasound. The company displayed conceptual imaging in development in GE Healthcare at ASE 2016 and at the American College of Cardiology 2017. The companies announced at ASE they now have a formal co-development agreement to make true 3-D imaging available for echo in the near future.
EchoPixel says it is working with a display vendor for 3-D screens that will eliminate the need to wear special glasses. The EchoPixal technology currently is only available as post processing, so it cannot be used for live procedures, but that may change as the two companies work together. If live, true 3-D transesophageal echo (TEE) imaging were developed, it could pave the way to eliminating the need for X-ray angiography to guide transcatheter structural heart procedures.
• Vector Blood Flow and Shear Stress Imaging: Vector flow imaging offers very detailed tracking of the flow of blood inside the heart and arteries, allowing visualization of speed of the blood in various anatomy, how it is influenced by atherosclerosis, stenotic valves, structural changes in the heart, or the implantation of devices. It is believed disruption of blood flow causing turbulence or the formation for swirling vortices play a role in disease development and may one day offer risk assessments of heart or vascular conditions long before the onset of symptoms. Vector flow imaging also can help quantify the amount of wasted energy the heart uses when it is pumping inefficiently, which may aid quantiftative analysis or risk predictions for heart failure.
Until now, vector flow imaging has only been a research tool and has not been available on any of the premium echocardiography imaging systems. Hitachi has offered vector flow imaging for several years. but not on a high-end echocardiography system until now. The company released its first premium cardiac ultrasound system at ASE, the Lisendo 880. In addition to offering comparable 2-D and 3-D image quality to the existing high-end, dedicated echo systems on the market, it also comes standard with vector flow imaging.
GE Healthcare also unveiled its prototype vector flow imaging technology at a lunch symposia at ASE 2017. Clinical examples of how it works and what it reveals were shown on the work-in-progress technology.
Hitachi's technology uses mathematical assumptions for calculating the vectors. GE’s system uses blood speckle tracking, which combines speckle tracking and high frame rates to track the echo returns of individual blood cells frame-by-frame. The Hitachi system takes about 30 seconds to a minute to process an image, and the GE system is supposed to process images in 10-20 seconds.
• Artificial Intelligence to Aid Reproducibility: Siemens Healthcare was the first to introduce elements of artifical intelligence (AI) algorithms into its cardiac echo systems several years ago to speed automation. Philips Healthcare also introduce elements of AI on its Epiq ultrasound system a couple years ago. It takes a 3-D echo dataset acquisition and automatically analyzes the image to identify the heart’s anatomy, labels it and then slices the optimal standard views for presentation. This eliminates issues with interoperator variability, because the software will always choose the optimal views based on machine learning, which uses thousands of prior exams representing the spectrum of patient anatomical variations. This would take years for a human operator to accumulate the same knowledge. Other vendors also have introduced elements of deep learning algorithms to help analyze echocardiograms or perform auto quantifications. Next generation echo systems will incorporate more AI features to further improve workflow by auto-completing time-consuming tasks and augmenting the sonographer so they can become more efficient and consistently accurate.
All the major imaging system vendors are either developing their own AI or partnering with AI vendors. Siemens Healthineers announced a partnership with IBM Watson earlier this year at HIMSS 2017. GE Healthcare announced in May it is working with Partners HeathCare, which will be executed through the newly formed Massachusetts General Hospital and Brigham and Women’s Hospital Center for Clinical Data Science. Mass General has been developing its own AI system for radiology. In addition to its Epic echo software, Philips also developed its own AI to support enhance its IntelliSpace Enterprise medical imaging informatics platform, which is smart enough to pull all the patient’s relevant prior exams for the same anatomy and open the images in the exact same view as the current exam.
Watch the VIDEO “Ultrasound's Integration of Artificial Intelligence and Robotic Echo,” a discussion with Partho Sengupta, M.D., professor of medicine, director of cardiovascular imaging and chair of cardiac innovations at the West Virginia University Heart and Vascular Institute, at ASE 2017.
Read the article, “How Artificial Intelligence Will Change Medical Imaging.”
Read the article “Use of Artificial Intelligence to Locate Standard Echo Heart Views.”
• Strain Imaging: Strain imaging, also referred to as speckle tracking and wall motion tracking, has gone from an interesting feature that few centers used or had on their echo systems, to a technology seeing rapid adoption. It has been around for years, but is now finally seeing wide spread adoption and will likely become part of the standard quantifications done in echo exams. It has the ability to better assess several cardiac conditions, monitor cardio-toxicity in oncology patients and aid in optimization of cardiac resychonization therapy (CRT). It also conduct qualification in a reliable, reproducible way, at least as long as all a patient's exams are all performed on the same vendor's machine, since the measures may vary between vendors as there is no standardization for how strain calculations are performed. Strain was discussed in all the sessions I attended and was a key point of vendor demonstrations on the expo floor.
Watch the VIDEO "CRT Optimization Using Echo," an interview with Leyla Elif Sade, M.D., MESC, professor of cardiology at Başkent University, Ankara, Turkey, on the use of echo for CRT optimization at the 2017 American Society of Echocardiography (ASE) annual meeting.
For more information on strain imaging, artificial intelligence and other advances in ultrasound, watch the VIDEO “Ultrasound Technology Trends and Technology to Watch.” It includes a discussion with Chi-Ming Chow, M.D., MSc, FRCPC, FACC, FASE, attending staff cardiologist, St. Michael's Hospital, and associate professor, Department of Medicine, University of Toronto, at ASE 2017.



 May 04, 2026
May 04, 2026