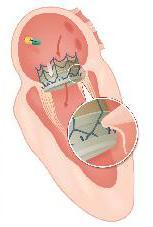
May 30, 2014 — CardiAQ Valve Technologies announced it successfully implanted its second-generation transcatheter mitral valve (TMVI) using the company’s newly added transapical delivery system. CardiAQ previously performed the first ever transfemoral transcatheter mitral valve implantation procedure in June 2012 with its self-conforming, self-anchoring implant technology. The transapical TMVI procedure was performed on an 88-year-old female suffering from severe mitral regurgitation (MR 4+) at The Heart Centre, Rigshospitalet University Hospital, Copenhagen, Denmark, by interventional cardiologists Lars Søndergaard, M.D., echocardiographer Nikolaj Ihlemann, M.D., cardiothoracic surgeons Peter Skov Olsen, M.D., Susanne Holme, M.D., and Arshad Quadri, M.D., and anesthesiologist Peter Bo Hansen, M.D.
“We are grateful to have had the opportunity to work with the CardiAQ technology again in order to provide treatment to this patient for whom no other alternatives were available,” said Søndergaard. “Based on a number of risk and anatomical factors, neither traditional mitral valve surgery nor transcatheter repair with MitraClip were options for this patient. Through careful review and consideration, we determined that TMVI, using CardiAQ’s second-generation transcatheter mitral valve and its new transapical delivery system, could offer this patient a chance at a longer and more comfortable life. The valve was implanted successfully with excellent function. The patient is recovering uneventfully and already feels a marked reduction in symptoms.”
“CardiAQ’s second-generation transcatheter mitral valve builds upon the company’s proprietary method for anchoring the implant through leaflet engagement, chordal preservation and annular attachment, while offering greater durability, improved flow properties and a novel feature for the prevention of paravalvular leaks,” said Arshad Quadri, M.D., founder and CMO of CardiAQ Valve Technologies.
“In the past few years, the potential of TMVI has become increasingly clear. As the first company to utilize the subvalvular apparatus in our attachment method, CardiAQ has set the standard for TMV anchoring,” said Brent Ratz, co-founder, president and COO of CardiAQ. “With the benefit of our first-generation first-in-human experience in 2012, we have gone on to further optimize our implant, incorporating all that we have learned into our second-generation TMV technology.”
For more information: www.cardiaq.com


 April 24, 2026
April 24, 2026 









