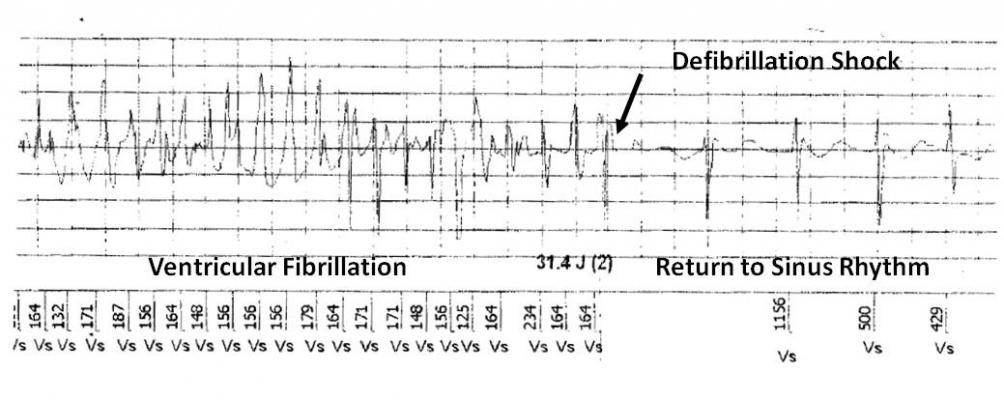
Figure 2. ICD electrogram showing successful defibrillation shock.
Sudden cardiac arrest (SCA) is a leading cause of mortality in the United States, accounting for an estimated 300,000 deaths each year – more than lung cancer, breast cancer and HIV/AIDS combined. Current risk stratification paradigms for SCA are designed to identify high?risk populations based primarily on assessment of left ventricular ejection fraction (LVEF). However, determining the risk for an individual patient remains a significant clinical challenge, particularly since most cardiac arrests occur in those without severe LV dysfunction [1, 2]. Microvolt T-wave alternans (MTWA) testing is a noninvasive tool that may be used to help identify underlying vulnerability to life-threatening arrhythmias [3-5]. This case presents a patient with hypertensive cardiomyopathy and moderate LV dysfunction who experienced cardiac arrest within 18 months of a positive MTWA test.
Case History
An asymptomatic 57-year-old male underwent a myocardial perfusion imaging (MPI) study to evaluate progression of hypertensive cardiomyopathy and previously documented severe LV dysfunction which had improved after aggressive treatment with carvedilol. A 12-lead ECG showed normal sinus rhythm with left bundle branch block (LBBB) morphology. Results of the adenosine SPECT MPI were abnormal revealing dilated cardiomyopathy (DCM) with evidence of left ventricular hypertrophy and an LVEF of 44 percent. A small apical perfusion deficit was also noted. Based on reduced LV function and DCM, a MTWA test was ordered to further clarify his risk for ventricular tachyarrhythmias and SCA.
MTWA Study and Follow-up
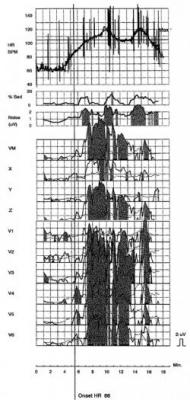
In November 2006, MTWA testing was performed with treadmill exercise using the analytic spectral method to measure alternans (HearTwave II, Cambridge Heart, Tewksbury, Mass.). At the time of the test, the patient was in normal sinus rhythm with complete LBBB and intermittent ectopy. The MTWA result was positive with an onset heart rate of 86 bpm (Fig. 1). The positive MTWA study, along with the patient’s other risk factors, prompted referral to an electrophysiologist (EP) who performed an EP study in December. During the procedure, ventricular tachycardia was readily induced and a single-chamber implantable cardioverter defibrilator (ICD — from Ovatio VR, ELA, Montrouge, France) was implanted. In April 2008, while at his home in Haiti, the patient experienced an episode of ventricular fibrillation that was appropriately treated with a 31.4 J defibrillation shock (Fig. 2).
Discussion
This case presents a patient with several clinical factors independently associated with increased risk of SCA including hypertension [6], dilated cardiomyopathy [7], left ventricular hypertrophy [8] and left bundle branch block [9]. While the patient was being aggressively treated for hypertensive cardiomyopathy and had shown improved systolic function, data suggests that mortality risk remains high [10]. Interventricular conduction delay likely worsens the prognosis as noted by Morin et al who found prolonged QRS duration to be independently associated with increased risk for SCA in hypertensive patients with ECG evidence of left ventricular hypertropy (LVH) undergoing intensive antihypertensive therapy [11].
MTWA has been cited as a useful adjunct tool in such patients with moderate LVEF for whom SCA is a “real but difficult-to-ascertain risk” [12]. In the case of nonischemic dilated cardiomyopathy, an abnormal TWA test is associated with increased risk of cardiac death and life-threatening arrhythmias in patients with moderate to severe LV dysfunction [4, 13, 14]. Kitamura et al found that the onset heart rate (OHR) for MTWA in DCM patients provides additional prognostic information; in multivariate analysis, an OHR less than 100 bpm was a more powerful predictor of arrhythmic events than LVEF or alternans alone [15].
In this case, the positive MTWA test with low onset HR helped identify the patient’s latent arrhythmic risk and was the trigger for further evaluation which ultimately led to life-saving therapy. Since the majority of sudden cardiac arrests occur in patients without severe left ventricular dysfunction, it is important to have a tool that can distinguish this risk.
References:
1. Myerburg RJ, et al. "Sudden cardiac death caused by coronary heart disease." Circulation 2012; 125(8): 1043-52.
2. Stecker EC, et al. "Population-based analysis of sudden cardiac death with and without left ventricular systolic dysfunction: two-year findings from the Oregon Sudden Unexpected Death Study." J Am Coll Cardiol 2006; 47(6): 1161-6.
3. Chow T, et al. "Prognostic utility of microvolt T-wave alternans in risk stratification of patients with ischemic cardiomyopathy." J Am Coll Cardiol 2006; 47(9): 1820-7.
4. Salerno-Uriarte JA, et al. "Prognostic Value of T-Wave Alternans in Patients With Heart Failure Due to Nonischemic Cardiomyopathy: Results of the ALPHA Study." J Am Coll Cardiol 2007; 50(19): 1896-1904.
5. Bloomfield DM, et al. "Microvolt T-wave alternans and the risk of death or sustained ventricular arrhythmias in patients with left ventricular dysfunction." J Am Coll Cardiol 2006; 47(2): 456-63.
6. Kannel WB, et al. "Hypertension, antihypertensive treatment, and sudden coronary death. The Framingham Study." Hypertension 1988; 11(3 Pt 2): II45-50.
7. Zipes DP, et al. Sudden cardiac death. Circulation 1998; 98(21): 2334-51.
8. Haider AW, et al. Increased left ventricular mass and hypertrophy are associated with increased risk for sudden death. J Am Coll Cardiol 1998; 32(5): 1454-9.
9. Zimetbaum PJ, et al. Electrocardiographic predictors of arrhythmic death and total mortality in the multicenter unsustained tachycardia trial. Circulation 2004; 110(7): 766-9.
10. de Carvalho Frimm C, et al. The long-term outcome of patients with hypertensive cardiomyopathy. J Hum Hypertens 2005; 19(5): 393-400.
11. Morin DP, et al. "QRS duration predicts sudden cardiac death in hypertensive patients undergoing intensive medical therapy: the LIFE study." Eur Heart J 2009; 30(23): 2908-14.
12. Mirro MJ. "Strategies for reducing sudden cardiac death: Application of microvolt T-wave alternans testing in clinical practice." Heart Rhythm 2009; 6(3): S45-S48.
13. Hennersdorf MG, et al. "T-wave alternans as a risk predictor in patients with cardiomyopathy and mild-to-moderate heart failure." Pacing Clin Electrophysiol 2000; 23(9): 1386-91.
14. Baravelli M, et al. "Predictive significance for sudden death of microvolt-level T wave alternans in New York Heart Association class II congestive heart failure patients: a prospective study." Int J Cardiol 2005; 105(1): 53-7.
15. Kitamura H, et al. "Onset heart rate of microvolt-level T-wave alternans provides clinical and prognostic value in nonischemic dilated cardiomyopathy." J Am Coll Cardiol 2002; 39(2): 295-300.


 May 08, 2026
May 08, 2026 









