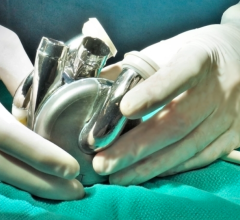
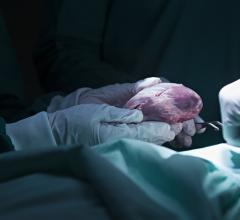
February 4, 2008 - The Centers for Medicare & Medicaid Services (CMS) announced that after completing a review of its 1986 national non-coverage policy for artificial hearts, its preliminary decision is to provide reimbursement for patients who receive the CardioWest temporary Total Artificial Heart (TAH-t) as part of FDA studies that meet CMS specifications.
Following a public comment period, CMS will post its final decision on May 1, 2008. "Our proposal relaxes a long-standing non-coverage policy, gives access to our beneficiaries and promotes evidence development through FDA approved studies of this advanced technology," said CMS Acting Administrator Kerry Weems. "This device may be able to help patients that otherwise have no treatment options available to them."
"This decision is a historic step toward making the artificial heart available to most Americans," explained Rodger Ford, president and CEO of SynCardia Systems, Inc., manufacturer of the CardioWest artificial heart. "Many smaller insurers use CMS reimbursement decisions as their benchmark for coverage."
The CardioWest artificial heart is currently covered by about half of insurers, including Aetna and BlueCross BlueShield. With final CMS approval, most insurers are likely to cover the CardioWest artificial heart.
On Aug. 1, 2007, CMS posted a notice to its website announcing the review of this non-coverage policy, after SynCardia submitted a formal request for coverage of the CardioWest artificial heart when used in accordance with its FDA-approved labeling as a bridge to human heart transplant.
The CardioWest artificial heart received FDA approval on Oct. 15, 2004. It is the world�s only FDA, CE and Health Canada approved temporary total artificial heart. It has been implanted in more than 700 patients, accounting for more than 120 patient years of life on the device.
For more information: www.cardiowest.com


 April 28, 2026
April 28, 2026