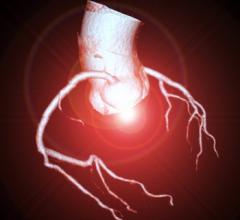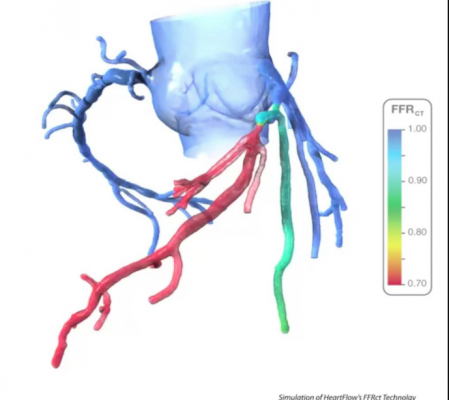
HeartFlow's FFR-CT analysis software uses a computed tomography scan and supercomputing fluid dynamics software to create a noninvasive, virtual FFR map of the entire coronary artery tree to determine the flow-limiting severity of ant lesions.
November 6, 2017 – The Centers for Medicare & Medicaid Services (CMS) has finalized a New Technology Ambulatory Payment Classification (APC) for the HeartFlow FFR-CT Analysis, a first-of-its-kind noninvasive technology that helps clinicians diagnose and treat patients with suspected coronary artery disease (CAD). Under the APC payment system, hospitals enrolled in Medicare that bill CMS for the HeartFlow FFR-CT Analysis for Medicare patients will be eligible for reimbursement at a rate of $1,450.50 for the technical component of the test. This payment rate will take effect on Jan. 1, 2018.
"The CMS decision to assign a new technology APC for the HeartFlow FFR-CT Analysis is a major milestone and will ensure that our innovative, noninvasive technology can be utilized for Medicare beneficiaries. This is a significant development, given the high prevalence of CAD in people over age 65,” said John Stevens, M.D., president and chief executive officer of HeartFlow. “This decision is a testament to the wealth of clinical evidence and cost data that support the use of the HeartFlow FFR-CT Analysis as a critical, potentially life-saving technology.”
CAD, which today affects 16.8 million Americans,[1] develops when the coronary arteries narrow, reducing blood flow to the heart and causing angina (chest pain), myocardial infarction (heart attack) and death. Older individuals are at highest risk of CAD.[2] The HeartFlow FFR-CT Analysis provides insight into both the extent of CAD and the impact of the disease on blood flow to the heart. It uses high-quality images from a coronary computed tomography (CT) angiogram to provide clinicians with functional information in order to offer actionable information on the optimal course of treatment.
According to CMS, more than 55 million Americans are enrolled in the Medicare program.[3]
“CMS’s decision to assign a new technology APC for FFR_CT technology for Medicare recipients is a recognition of the value of this technology and its demonstrated ability to reduce the number of invasive diagnostic coronary angiography procedures and help medical centers reduce costs,” said Pamela Douglas, M.D., Ursula Geller Professor for Research in Cardiovascular Disease in the School of Medicine at Duke University and past president of the American College of Cardiology. “The HeartFlow Analysis is an important tool in helping us assess patients with suspected coronary artery disease and better understand how their coronary blockages affect blood flow to the heart.”
“The decision by CMS will help in the diagnosis and treatment of patients with suspected CAD and means the number of patients over age 65 who will have access to this technology will significantly increase,” said Daniel Simon, M.D., president, University Hospitals (UH) Cleveland Medical Center, and professor of medicine at Case Western Reserve University School of Medicine. “At our center, the use of the HeartFlow FFR-CT Analysis is transforming how we diagnose and treat patients with coronary artery disease, helping us move closer to achieving the triple aim of improving the patient experience, improving the health of our patients, and reducing healthcare costs at our institution.”
Previously, the American Medical Association (AMA) issued a set of Category III Current Procedural Terminology (CPT) codes for the HeartFlow FFR-CT Analysis. These codes provide evidence that the Analysis is a unique and distinct procedure that provides important and actionable clinical information above and beyond that of a coronary CT angiogram. Beginning on Jan. 1, 2018, hospitals utilizing FFR-CT will be able to generate claims when it is ordered for patients with suspected CAD via the use of specific CPT codes.
The CMS decision follows a growing number of commercial payers and professional organizations that have recognized its value in diagnosing patients with suspected CAD. Anthem, Aetna and other Blue Cross Blue Shield companies that collectively provide health insurance for approximately 80 million people have issued positive medical coverage decisions. Evidence Street, which conducts healthcare technology evaluations for the Blue Cross Blue Shield Association, a national federation of 36 independent Blue Cross and Blue Shield companies, issued a positive review. In the U.K., the National Institute for Health and Care Excellence (NICE) of the National Health Service, which covers 64.6 million lives, also issued positive guidance. The American College of Cardiology (ACC) and American Heart Association (AHA) released updated Appropriate Use Criteria for Coronary Revascularization in Patients with Stable Ischemic Heart Disease. These criteria include the use of HeartFlow FFR-CT Analysis in determining the appropriateness of revascularization in many clinical scenarios.
With HeartFlow’s technology, data from a patient’s coronary CT angiogram are securely uploaded from the hospital’s system to the cloud. HeartFlow leverages deep learning to create a personalized, digital 3D model of the patient’s coronary arteries. It then uses powerful computer algorithms to solve millions of complex equations to simulate blood flow and assess the impact of blockages on coronary blood flow. The FFR-CT Analysis is provided to the patient’s physician via a secure web interface.
For more information: www.heartflow.com
Related FFR-CT articles and Videos
Clinical Applications of FFR-CT
VIDEO: Early U.S. Experience With FFR-CT in Evaluating ED Chest Pain Presentation
FFR-CT Shown to Reduce Costs in Patients with Chest Pain
VIDEO: New CPT Reimbursement Codes for Cardiology
FFR-CT Gains CPT Code for Reimbursement
VIDEO: CT-FFR to Noninvasively Assess Coronary Lesions
References:
1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2787400/.
2. https://www.nhlbi.nih.gov/health/health-topics/topics/cad/atrisk.
3. https://www.cms.gov/Newsroom/MediaReleaseDatabase/Press-releases/2015-Press-releases-items/2015-07-28.html.
4. Douglas PS, DeBruyne B, Pontone G., Patel MR, et al. One-year outcomes of FFRCT-guided care in patients with suspected coronary disease: The PLATFORM Study. J Am Coll Cardiol. 2016;68(5),435-45.

 February 06, 2026
February 06, 2026