May 24, 2022 — Anumana, Inc., an AI-driven health technology company from nference, Inc., today announced that the U.S. Food & Drug Administration (FDA) has granted Breakthrough Device Designation to its AI-enhanced, ECG-based Pulmonary Hypertension (PH) Early Detection Algorithm. The algorithm is a precise, non-invasive screening tool that addresses an unmet need for earlier diagnosis of patients with pulmonary hypertension, which may otherwise go unnoticed until the disease has advanced, delaying treatment initiation, thereby limiting treatment efficacy and adversely impacting patient outcomes.
FDA Breakthrough Device Designation recognizes novel innovations that demonstrate the potential to provide more effective treatment or diagnosis for life-threatening or irreversibly debilitating diseases and helps to accelerate the regulatory process.
“Electrophysiology waveforms hold immense untapped potential for detecting diseases earlier in their natural history, particularly for conditions in which earlier diagnosis and therapeutic intervention can prolong survival and improve quality of life,” said Venky Soundararajan, PhD, co-founder and Chief Scientific Officer of Anumana and nference. “The FDA's Breakthrough Device Designation for Anumana's PH Early Detection Algorithm is one step forward for the field of ECG AI overall, and more saliently, a giant leap forward for PH patients.”
PH is a progressive, life-threatening disease estimated to affect up to 0.1-1% of the global population.1,2 Due to the non-specific symptoms of the disease, such as shortness of breath, diagnostic delays are common3,4,5, often longer than a year, and have been linked with a higher mortality risk in certain patient subgroups.6,7
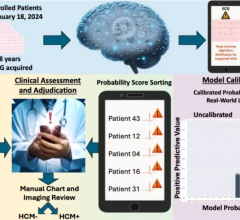
Anumana’s AI-enhanced ECG algorithm is designed to easily and safely detect PH at an earlier stage, by leveraging the widespread availability of 12-lead ECGs in primary care, urgent care, and emergency room settings. The AI-enhanced ECG PH Algorithm is enabled by the nference platform, which provides insight from more than 6 million de-identified patient records, including over 8 million ECGs. It was developed through a collaboration between data scientists and physicians at Anumana, Janssen Research & Development, LLC, and Mayo Clinic. Supported by Mayo Clinic Platform, nference launched Anumana in 2021 to develop and commercialize AI-enabled algorithms.
If approved, the early detection algorithm will be offered as Software as a Medical Device (SaMD) that can be downloaded on a physician’s smartphone, tablet, or computer, or accessed via the Cloud through an Electronic Health Record or ECG Information Management System interface. Using a standard 12-lead ECG, the algorithm analyzes the voltage-time data and within seconds provides a prediction of likelihood of PH, reducing the time between initial symptoms and the first evaluation for PH via targeted cardiac imaging. This early detection algorithm is still in development and not yet approved for clinical use.
“While therapeutic options for patients with pulmonary hypertension have evolved in recent years, we have not seen significant advancement in reducing the time from symptom onset to diagnosis – and our hypothesis was that data science could help change this,” said Najat Khan, PhD, Chief Data Science Officer and Global Head, Strategy and Operations, Janssen Research & Development, LLC. “We leveraged the power of artificial intelligence, the ingenuity of our data scientists and researchers, and deep collaboration to co-develop this AI-based innovation, with the ultimate goal of helping to improve patient outcomes and transform the trajectory of this devastating disease.”
“Early diagnosis of pulmonary hypertension is paramount due to its progression and potential severity,” said Paul Friedman, M.D., chair of the Department of Cardiovascular Medicine at Mayo Clinic. “The addition of AI to a standard ECG – a painless, inexpensive, widely used test that is routinely performed – transforms the ECG into a screening tool for PH, with the opportunity to improve outcomes via early detection by guiding appropriate testing.”
The Pulmonary Hypertension Early Detection Algorithm is the company’s second technology to receive FDA Breakthrough Device Designation. Its low ejection fraction algorithm, developed by Mayo Clinic and licensed by Anumana, received Breakthrough Device Designation in 2019 and Emergency Use Authorization for COVID-19 in 2020. Anumana has a large pipeline of novel algorithms that have been validated by over 30 peer-reviewed publications, including a first of its kind prospective clinical impact study on low ejection fraction that was published in Nature Medicine in 2021. These software solutions are currently in development with each algorithm as a candidate for FDA medical device approval.
For more information: www.anumana.ai
References:
- Wijeratne DT, Lajkosz K, Brogly SB, et al. Increasing Incidence and Prevalence of World Health Organization Groups 1 to 4 Pulmonary Hypertension. Circ Cardiovasc Qual Outcomes. 2018;11(2):e003973. doi:10.1161/CIRCOUTCOMES.117.003973
- Hoeper MM, Humbert M, Souza R, et al. A global view of pulmonary hypertension. Lancet Respir Med. 2016;4(4):306-322. doi:10.1016/S2213-2600(15)00543-3
- Armstrong I, Billings C, Kiely DG, et al. The patient experience of pulmonary hypertension: a large cross-sectional study of UK patients. BMC Pulm Med 2019: 19: 67.
- Brown LM, Chen H, Halpern S, et al. Delay in recognition of pulmonary arterial hypertension: factors identified from the REVEAL Registry. Chest 2011: 140: 19-26.
- Rich S, Dantzker DR, Ayres SM, et al. Primary pulmonary hypertension. A national prospective study. Ann Intern Med 1987: 107: 216-223.
- Khou V, Anderson JJ, Strange G, et al. Diagnostic delay in pulmonary arterial hypertension: Insights from the Australian and New Zealand pulmonary hypertension registry. Respirol Carlton Vic. 2020;25(8):863-871. doi:10.1111/resp.13768
- Klok FA, Barco S, Konstantinides SV, et al. Determinants of diagnostic delay in chronic thromboembolic pulmonary hypertension: results from the European CTEPH Registry. Eur Respir J. 2018;52(6):1801687. doi:10.1183/13993003.01687-2018


 September 24, 2025
September 24, 2025