March 22, 2010 – An FDA committee voted unanimously in favor of approval with conditions of a new pacemaker that is safe for use in magnetic resonance imaging (MRI) systems. MRI procedures are not recommended in the United States for patients who currently have implanted pacemakers. If approved, Revo MRI pacemaker has the potential to be the first FDA-approved pacing system designed for use in the MRI setting.
The FDA Circulatory System Devices Panel of the Medical Devices Advisory Committee recommended Revo MRI for approval with conditions related to the planned post-market study, healthcare professional training, and labeling to reflect MRI scans are to be conducted with the full Revo MRI SureScan Pacing System. The FDA will consider the panel’s recommendation in its review of Revo MRI; however, it is not bound by its advisory committee’s recommendations.
MRI is critical in the diagnosis of many serious conditions, but patients with current pacemakers most often do not have access to this technology. It is estimated that more than 200,000 patients annually in the United States have to forego an MRI scan because they have a pacemaker (1) due to the risks involved, including interference with pacemaker operation, damage to system components, lead or pacemaker dislodgement, heating of the lead tips and unintended cardiac stimulation. (2, 3, 4, 5) MRI scans allow physicians to make a wide range of health diagnoses by viewing highly detailed images of internal organs, blood vessels, muscle, joints, tumors, areas of infection and more.(6)
The FDA panel reviewed safety and effectiveness data from a prospective, randomized multicenter trial at 42 centers around the world featuring 464 implanted patients. Major inclusion criteria included standard class I or II dual chamber pacemaker indication, which allowed for pacemaker-dependent patients. Patients were excluded if they had previously implanted medical devices or abandoned leads. Patients were randomized at implant to either receive an MRI or not to receive an MRI.
The primary endpoints evaluated were safety and effectiveness of the Revo MRI pacing system in the MRI environment. For safety, the MRI group was evaluated for MRI procedure-related complications through one month post MRI. The primary effectiveness endpoint tested equivalence between MRI versus control for atrial and ventricular pacing capture thresholds and atrial and ventricular sensed amplitudes through one month post MRI.
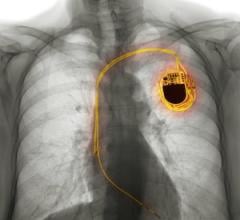
The device and leads also contain radiopaque markers, viewable via X-ray, to indicate that the device is MR-Conditional. MR-Conditional is a term used to indicate that a device may be used in the MRI environment under certain conditions, such as a particular type of MRI scanner and scanner settings.
For more information: www.medtronic.com
References:
1. Medtronic calculations cited in Rod Gimbel and Ted McKenna, “Safety of Implantable Pacemakers and Cardioverter Defibrillators in the Magnetic Resonance Imaging Environment,” Business Briefing: Long-Term Healthcare 2005, available at www.touchbriefings.com.
2. Faris OP, Shein M. “Food and Drug Administration perspective: Magnetic resonance imaging of pacemaker and implantable cardioverter-defibrillator patients.” Circulation 2006, 114:1232-1233.
3. Roguin A, Schwitter J, Vahlhaus C, et al. “Magnetic resonance imaging in individuals with cardiovascular implantable electronic devices.” Europace 2008, 10:336-346.
4. Levine GN, Gomes AS, Arai AE, et al. “Safety of magnetic resonance imaging in patients with cardiovascular devices: an American Heart Association scientific statement from the Committee on Diagnostic and Interventional Cardiac Catheterization, Council on Clinical Cardiology, and the Council on Cardiovascular Radiology and Intervention: endorsed by the American College of Cardiology Foundation, the North American Society for Cardiac Imaging, and the Society for Cardiovascular Magnetic Resonance.” Circulation 2007, 116:2878-2891.
5. Kalin R and Stanton MS. “Current clinical issues for MRI scanning of pacemaker and defibrillator patients.” PACE 2005, 28:326-328.
6. American College of Radiology (ACR) and the Radiological Society of North America (RSNA). “MRI of the body (chest, abdomen, pelvis).” June 10, 2009. Available at http://www.radiologyinfo.org/en/info.cfm?pg=bodymr. Accessed March 11, 2010.

 April 24, 2026
April 24, 2026 









