
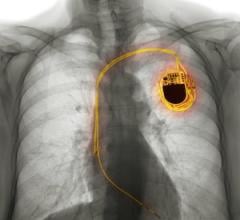
April 17, 2012 — St. Jude Medical Inc. announced the first implant in its Accent MRI Pacemaker and Tendril MRI Lead investigational device exemption (IDE) study. The ultimate goal of the study is to determine if patients with these devices can safely undergo full-body, high-resolution magnetic resonance imaging (MRI) scans to better accommodate their medical needs. The investigational Accent MRI Pacemaker system offers an advanced pacing platform that provides wireless telemetry and algorithms to help address individual patient conditions.
MRI scans provide imaging information to help evaluate the presence of certain diseases or conditions that may or may not be adequately assessed using other imaging methods, such as X-ray or ultrasound. Because traditional pacing systems may be affected during an MRI scan, patients are currently discouraged from receiving the scans. Approximately 1 million people are implanted with pacemakers worldwide, and approximately 300,000 of those patients could benefit from scans of the major organs and bones in the thoracic region of the body during the lifetime of their devices.
“Because young pacemaker patients have a high likelihood of needing an MRI over their lifetime, and older pacemaker patients often have co-morbidities, they may have other conditions, which could benefit from MRI scans of internal organs or major bones and in particular may benefit from cardiac MRI scans,” said Raymond Schaerf with Providence Saint Joseph Medical Center in Burbank, Calif., who implanted the first pacemaker in the study. “Currently, there is no pacemaker available that allows for full-body MRI scans, which further assists physicians in the early diagnosis and treatment of certain diseases, such as cancer or stroke, as well as other medical conditions facing heart patients.”
The Accent MRI Pacemaker and Tendril MRI Lead study, conducted under an investigational device exemption (IDE) from the U.S. Food and Drug Administration (FDA), is a randomized, clinical trial assessing the safety and efficacy of the Accent MRI system in patients with standard bradycardia (a heart rate that is too slow) who are indicated for a pacemaker. The clinical trial will take place at a maximum of 80 centers worldwide and will enroll approximately 800 patients. All patients in the study will be implanted with an Accent MRI Pacemaker and a Tendril MRI lead and be followed for 12 months post-implant to evaluate the acute and chronic performance of the system. A subset of patients will be randomized to receive an MRI after enrollment in the study.
The study also intends to investigate a unique solution developed by St. Jude Medical that streamlines the workflow at both the cardiology center and the radiology center. The SJM MRI Activator is a handheld device that offers a simple option for programming the Accent MRI Pacemaker to the appropriate MRI mode for use during the MRI scan. By pushing a single button, the SJM MRI Activator can program an Accent MRI Pacemaker’s pacing system into an MRI-scan safe mode and restore it back to the original setting once the scan is completed. The settings can be established during routine device follow-up visits before the MRI scan takes place using the Merlin Patient Care System programmer.
The Accent MRI pacemaker is based on the company’s latest Accent RF Pacemaker platform and has advanced features designed to improve patient care. Wireless communication, available with the Accent MRI RF model, streamlines the device implant procedure and follow-up appointments, and enables more consistent home monitoring by eliminating the need for any patient interaction with automated daily device checks, rather than requiring the patient to keep a telemetry wand positioned over the device.
The Tendril MRI Lead body has a co-axial design and Optim outer insulation, a silicone-polyurethane co-polymer material, which offers increased durability, improved flexibility and contributes additional handling characteristics and control during implant procedures.
For more information: sjm.com

 April 24, 2026
April 24, 2026 









