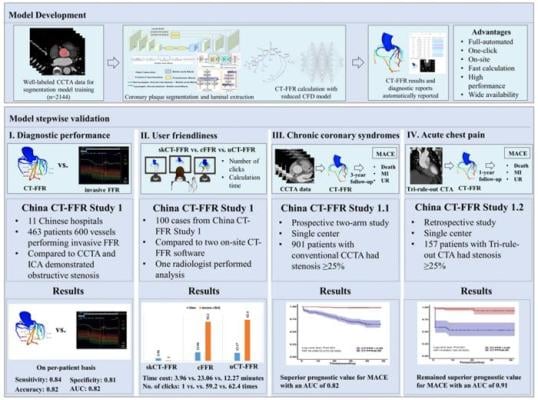
Study design and brief results of this study. ©Science China Press
May 1, 2024 —In this 3-stage study, 3 cohorts were used for diagnostic performance, and prognostic performance evaluation for this fully automated, one-click, on-site CT-FFR technique. This study was led by Professors Long Jiang Zhang and Guang Ming Lu (Department of Radiology, Jinling Hospital, Affiliated Hospital of Medical School, Nanjing University).
In cohort 1, a total of 463 patients and 600 coronary vessels with CCTA images and following invasive fractional flow reserve (FFR) results were collected for diagnostic performance and user-friendliness evaluation. CT-FFR had a strong correlation with invasive FFR both on a per-vessel basis and per-patient basis (Pearson R = 0.68, 95% CI: 0.63–0.72 and Pearson R = 0.70, 95% CI: 0.65–0.75, P <0.0001, respectively). CT-FFR had an sensitivity of 0.84, specificity of 0.81, and accuracy of 0.82 for identifying functional myocardial ischemia on a per-patient basis. On a per-vessel basis, sensitivity, specificity, and accuracy of CT-FFR were 0.80, 0.83, and 0.82, respectively. Furthermore, the authors have compared the user-friendliness between this fully-automatic on-site CT-FFR with other two manual-dependent on-site CT-FFR software (Siemens, cFFR and United-Imaging, uCT-FFR), and found it was associated with significantly reduced calculation time (less than 4 minutes) and number of mouse clicks (only one-click) of CT-FFR analysis.
In cohorts 2 and 3, a total of 901 participants with chronic coronary syndromes and 157 participants with acute chest pain were collected, respectively. Worse outcomes were associated with positive CT-FFR results (CT-FFR ≤ 0.80) than negative CT-FFR results (CT-FFR > 0.80) both in participants with chronic coronary syndromes and ones with acute chest pain.
This is the first reported fully-automatic CT-FFR technique, and demonstrated similar diagnostic performance compared to HeartFlow and Siemens CT-FFR technique.
For more information: https://www.sciencedirect.com/


 August 29, 2025
August 29, 2025