News | April 08, 2014
GE Healthcare Debuts Connected Cardiovascular Care at ACC 2014
New cardiology offerings aimed at safe, fast and accurate imaging and treatment of the heart
April 7, 2014 — GE Healthcare showcased a new suite of connected cardiovascular technologies, including several for advanced imaging and new software capabilities, at ACC 2014. New innovations included advances in molecular diagnostics, super-speed computed tomography (CT) scans, and patient-friendly electrocardiograms.
Life Sciences
GE Healthcare’s molecular imaging agent AdreView (Iobenguane I-123) is the only FDA-approved molecular imaging agent for scintigraphic assessment of sympathetic innervation of the heart that can be used to help identify adult patients with lower one and two year mortality risk. The heart is supplied with sympathetic nerves that control cardiac conduction. Increased sympathetic nerve activity depletes norepinephrine (NE) storage and can signal heart failure, which may lead to cardiac death. AdreView imaging helps assess NE levels via an external imaging gamma camera. Serious hypersensitivity reactions have been reported following AdreView administration. The most common adverse reactions in NDA clinical trials — dizziness, rash, pruritus, flushing, headache, and injection-site reactions — occurred in less than 1.3 percent of patients.
Healthcare IT
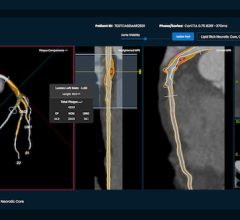
With Centricity Cardio Enterprise solution, cardiologists have a single point of access to unified patient data, images and reports across multiple modalities with end-to-end configurable workflows. The solution helps enhance patient care and financial performance by providing full clinical access, improved productivity and revenue cycle efficiencies.
The Centricity Cardio Enterprise solution comprises Centricity Cardio Imaging and Centricity Cardio Workflow. Centricity Cardio Imaging is a Web-based cardiovascular PACS that provides access to diagnostic quality images from anywhere with an Internet connection to help enable fast, more informed decisions. It helps optimize patient care and revenue cycle efficiencies by empowering users to manage the entire cardiovascular department, including scheduling, procedural data monitoring, report generation, billing and inventory management. At ACC, GE Healthcare introduced an expanded Centricity Cardio Workflow offering, including a new invasive peripheral vascular module to help cardiologists assess, document and report on the patient’s peripheral vessels.
Computed Tomography
GE Healthcare’s 510(k)-pending Revolution CT delivers uncompromised image quality and clinical capabilities through the convergence of coverage, spatial and temporal resolution in one system. Revolution also enables comprehensive cardiac exams with anatomic and functional information in just one heartbeat, even in challenging patients with higher heart rates.
Interventional Cardiology
Brent Gordon, M.D., of Loma Linda University utilizes GE’s Innova 3D and Innova HeartVision technologies to confidently manage complex procedures. “The Innova 3D and Innova HeartVision technologies, as well as cardiac analysis tools allow me to quickly define and analyze the anatomy of interest and strategize my intervention with each patient’s unique anatomy in mind,” said Gordon. “With a single injection, I can feel extremely confident in my measurements, determine optimal gantry angles, and create a virtual model that assists me in real-time through my planned intervention.”
Electrophysiology
Catheter ablation of atrial fibrillation is becoming an increasingly utilized therapy. A Duke Heart Center study of periprocedural imaging in catheter ablation involving more than 11,000 ablations, suggests this procedure’s outcomes improve when combined with adjunct imaging like CT, MR or ultrasound. According to the Duke study, when imaging is paired with atrial fibrillation catheter ablation, patients are at a lower risk for strokes and repeat ablation rates decrease.
“We found significant variation in how physicians image patients before and during AF ablation,” said Jonathan Piccini, M.D., assistant professor of medicine at Duke University Medical Center. “The periprocedural imaging was associated with improved outcomes. These findings are hypothesis generating, but highlight the importance of imaging decisions in catheter ablation. Significant opportunities exist to streamline these imaging technologies and to test best practices via prospective studies of multimodality imaging.”
Ultrasound
GE Healthcare ultrasound’s Vivid Cardiovascular Ultrasound systems provide innovative tools designed to help improve workflow efficiency through simplified image acquisition, intuitive navigation and advanced, yet easy to use quantification. New technologies showcased included the XDclear technology, Polar Vision and Depth Illumination communication enhancements.
Diagnostic Cardiology
The new MAC 2000 Resting ECG System with the latest version of Marquette 12SL analysis helps avoid variations in care delivery by standardizing ECG information from the physician office through the hospital. The system provides proven diagnostic support enabling clinicians to make fast, accurate diagnoses. Its flexible connectivity solutions offer clinicians the data they need to help improve patient outcomes and streamline workflow throughout the continuum of cardiac care.
The Muse ECG management system is now bi-directionally open to Non-GE electrocardiographs for improved care collaboration and asset management. Muse can now bridge a healthcare system’s ECG investments through bi-directional data sharing with both Non-GE and GE electrocardiographs. This solution offers comprehensive data access for clinicians by integrating ECG data from devices manufactured by vendors such as Philips, Mortara, Nihon Kohden, Edan, and Schiller in addition to GE.
The new app-driven Seer 1000 Holter Recorder from GE Healthcare utilizes innovative technology to establish patient and physician confidence right from the start. The Seer 1000 is a small, comfortable multi-channel digital Holter recorder paired with the Seer 1000 application for tablets, smartphones or Bluetooth enabled PCs. The system enables fast and accurate patient data entry, visual confirmation of lead placement and a clear view of waveform quality starting from initial hookup. The battery life indicator provides an assurance that battery life is adequate prior to beginning a test. One button operation makes it simple for patients to use, which may help reduce the number of repeat studies.
For more information: www.gehealthcare.com


 April 15, 2026
April 15, 2026