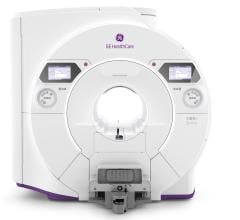
February 6, 2017 — At the 2016 Radiological Society of North America (RSNA) annual meeting, GE Healthcare unveiled Freelium, a magnet technology designed to use 1 percent of liquid helium compared to conventional magnetic resonance imaging (MRI) magnets. Instead of the average 2,000 liters of precious liquid helium, Freelium is designed to use only about 20 liters.
Helium is a critical component in MRI systems that has gone through two potential shortage crises, impacting hospitals and patients around the globe. But the helium supply is finite and demand has been rising over the past decades.
MRI uses superconducting magnets cooled to minus 452 degrees Fahrenheit in order to take high-definition pictures of a patient’s brain, vital organs or soft tissue. The only way to keep MRI magnets currently in clinical use that cold is by using thousands of liters of liquid helium mined from below the earth’s crust.
Magnets with Freelium technology are designed to be less dependent on helium, much easier to site, and eco-friendly. Thanks to Freelium technology, hospitals would no longer need extensive venting that often necessitates siting a magnet in a separate building or newly constructed room. Additionally, a Freelium magnet would not need any refilling during transportation nor throughout its lifetime. Therefore, when the Freelium technology is integrated into a commercialized product in the future, it could make MRI more accessible and less expensive to site and operate. This is particularly important in developing regions that lack necessary infrastructure, and in major metropolitan cities where siting a magnet can cost more than the magnet itself. Patients who currently do not have access to the diagnostic benefits of MRI today may have access in the future due to this breakthrough technology.
Freelium is technology in development that represents ongoing research and development efforts. This technology is not a product and may never become part of a product. It is not for sale and is not cleared or approved by the U.S. Food and Drug Administration (FDA) or any other global regulator for commercial availability.
For more information: www.gehealthcare.com


 May 14, 2026
May 14, 2026