July 24, 2009 – The 30-day survival improved in patients with acute myocardial infarction complicated by cardiogenic shock who were treated with intra-aortic balloon pump (IABP) counterpulsation, according to a new study in the American Journal of Cardiology (vol. 104, issue 3, pages 327-332, August 2009).
Although IABPs are increasingly being used for the treatment of patients with cardiogenic shock from acute myocardial infarction, there is little data on the long-term outcomes. Researchers at the Thoraxcenter, Erasmus Medical Center in Rotterdam, The Netherlands, and the Department of Intensive Care Medicine, Academic Medical Center, Amsterdam, The Netherlands, evaluated the 30-day and long-term mortality and to identify predictors for 30-day and long-term all-cause mortality of patients with AMI complicated by cardiogenic shock who were treated with an IABP.
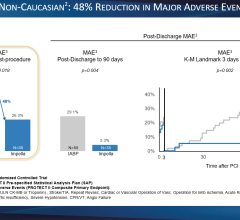
From January 1990 to June 2004, 300 consecutive patients treated with IABP were included. The mean age of the study population was 61 ± 11 years, and 79 percent of the patients were men. The survival rate until IABP removal after successful hemodynamic stabilization was 70 percent (n = 211). The overall cumulative 30-day survival rate was 58 percent. The 30-day mortality rate decreased over time from 52 percent in 1990 to 1994 to 36 percent in 2000 to 2004. Follow-up ranged from zero to 15 years. In patients who survived until IABP removal, the cumulative 1-, 5-, and 10-year survival rate was 69, 58 and 36 percent, respectively. The adjusted predictors of long-term mortality were arrhythmias during the intensive cardiac care unit stay (hazard ratio [HR] 1.8, 95 percent confidence interval [CI] 1.2 to 2.9) and renal failure during the intensive cardiac care unit stay (HR 2.5, 95 percent CI 1.3 to 5.1). After adjustment, treatment with primary percutaneous coronary intervention (HR 0.5, 95 percent CI 0.3 to 0.9) and coronary artery bypass grafting (HR 0.4, 95 percent CI 0.2 to 0.8) were associated with lower long-term mortality.
For more information: www.ajconline.org

 August 14, 2023
August 14, 2023