May 17, 2024 — Implicity, a leader in remote patient monitoring and cardiac data management solutions, announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for a new groundbreaking algorithm, SignalHF1, used in its remote monitoring solution.
Implicity was the first private company authorized to access the Health Data Hub, one of the largest databases of patients with heart disease in the world. Based on the Health Data Hub complete medico-administrative database and comprehensive datasets from Implicity's AI-based remote monitoring platform, the Implicity team trained and validated a machine learning algorithm that assesses the risk of future heart failure events, enabling physicians to intervene before a patient requires hospitalization.
"Heart failure remains a significant healthcare challenge, contributing to approximately one million hospitalizations each year. Preventing even a portion of these would be a game-changer in cardiac care," said Dr. Arnaud Rosier, Implicity's CEO and cardiologist. "SignalHF is an innovative and effective tool that physicians can use to assess risk of hospitalization due to heart failure earlier, enabling interventions that can lead to better outcomes and reduce hospital admissions."
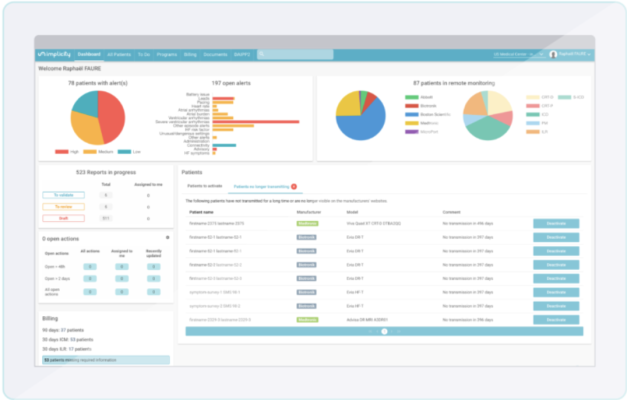
SignalHF is unique in that the manufacturer-agnostic solution can be used to analyze data collected from a variety of Cardiac Implantable Electronic Devices (CIED), including implantable cardiac defibrillators (ICD), pacemakers (PM) and cardiac resynchronization therapy devices (CRT-P, CRT-D) from Biotronik, Boston Scientific and Medtronic. SignalHF is the first pacemaker compatible heart failure algorithm on the market. The algorithm can accurately identify trends indicating whether a patient's condition is worsening. Care teams receive an automatic alert when the patient's risk score crosses an established threshold, indicating an individual is at high risk of experiencing heart failure and being hospitalized in the near future.
"Comprehensive heart failure management includes treatment, prevention, and personalization. Implicity's solution is part of this approach. Our alerts are generated in context with a patient's medical profile to help personalize care management plans. Furthermore, 75% of the alerts preceding a patient hospitalization are sent at least 14 days in advance – offering a two-week window to adjust medications or take proactive measures," said Issam Ibnouhsein, Head of Data at Implicity.
1 SignalHF is an FDA-cleared Class II medical device. See the instructions for use for more information.
For more information: www.implicity.com


 April 24, 2026
April 24, 2026