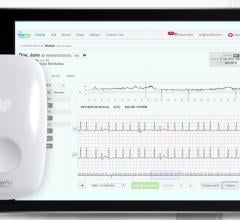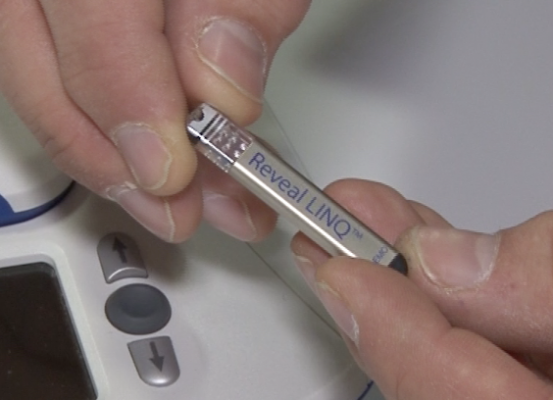
April 29, 2014 — John Muir Health announced that its Concord, Calif., medical center is one of the first hospitals in Northern California to implant a miniature cardiac monitor in a patient – the Reveal Linq Insertable Cardiac Monitor (ICM) System.
“This new device is part of a powerful system that detects and delivers continuous cardiac monitoring over long periods of time, giving physicians the ability to capture a more accurate picture of a patient’s heart rhythm,” said Susan Eisenberg, M.D., Cardiac Rhythm Center medical director at John Muir Health. “For patients who may be at risk of cardiac arrhythmias or experiencing symptoms of palpitations or dizziness, this device gives us an effective way to make an accurate diagnosis and determine an appropriate treatment plan.”
The Reveal Linq ICM, approved by the U.S. Food and Drug Administration (FDA) in February 2014, is for patients who experience symptoms such as dizziness, palpitation, fainting and chest pain that may suggest a cardiac arrhythmia, and for patients at increased risk for heart rhythm disorders. The symptoms may come and go making one-time tests such as electrocardiograms, less reliable than continuous monitoring. A 2011 study showed that patients with implantable long-term heart monitors who experienced arrhythmias received diagnoses much faster than non-implantable cardiac monitors; the time to diagnosis was reduced by 79 percent.
The Reveal Linq ICM is extremely small, approximately one-third the size of a AAA battery (roughly 1 cubic centimeter), making it more than 80 percent smaller than other ICMs. The device, made by Medtronic, allows physicians to continuously and wirelessly monitor a patient’s heart for up to three years. By contrast, external monitors typically are attached to the outside of a patient's body for monitoring from only one to 30 days.
The Linq ICM system also provides remote monitoring through the Carelink Network, which allows physicians to receive alerts about patients who are experiencing cardiac events. The Reveal Linq also includes the new MyCareLink Patient Monitor, a monitoring system using global cellular technology to transmit a patient’s diagnostic data to their clinician from nearly any location in the world.
“We are very excited to be able to offer patients the most technologically advanced device for heart monitoring,” said Eisenberg. “The size of the device also makes it nearly invisible on the body so patients can continue their normal activities without fear of detaching any wires or external monitors.”
The Reveal Linq is injected just beneath the skin through a small incision of less than 1 cm in the upper left side of the chest. The implant takes less than five minutes and requires no sedation. Patients are able to go home just minutes after the procedure. The Reveal Linq is also MR-conditional, which allows patients to undergo magnetic resonance imaging (MRI), if needed.
Eisenberg implanted the first Reveal Linq device in a patient at John Muir Health in the middle of March. In addition to the Reveal Linq, John Muir Health was one of the first Bay Area hospitals to implant the world’s first defibrillator with no electrodes for patients at risk for sudden cardiac arrest.
For more information: www.johnmuirhealth.com


 July 22, 2025
July 22, 2025