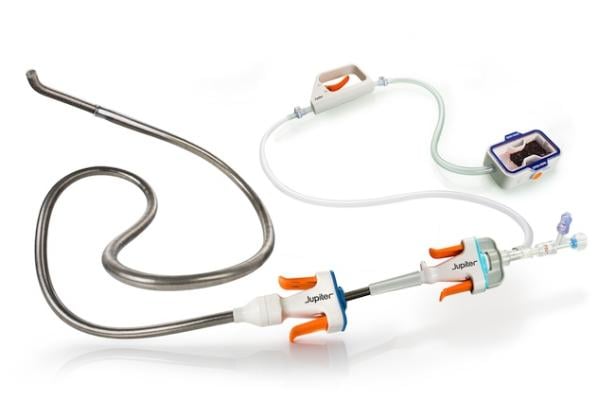
Photo: Jupiter Endovascular, Inc.,
Oct. 26, 2025 – Jupiter Endovascular, Inc., a medical technology company developing a new class of endovascular interventions using its Transforming Fixation (TFX) technology, announced positive results from the SPIRARE I first-in-human (FIH) study. Data was presented at the Transcatheter Cardiovascular Therapeutics (TCT 2025) conference by Professor Irene Lang, MD, Principal Investigator and Professor of Vascular Biology at the Medical University of Vienna in Austria.
SPIRARE I (NCT06571760) is the first study to evaluate the safety and performance of the Vertex Pulmonary Embolectomy System featuring TFX technology in patients with acute, intermediate-risk pulmonary embolism (PE). The prospective, multicenter European study enrolled 10 patients across two sites in Austria and Poland under the leadership of Professor Lang, MD and Professor Grzegorz Kopec, MD, in collaboration with Professor Krzysztof Bartus, MD.
“TFX represents an entirely new way to perform endovascular interventions, overcoming the long-standing tradeoff between device access and delivery,” said Professor Lang. “Pulmonary embolism remains an area of major unmet need — both in the acute phase and over the long term — where its sequelae may contribute to chronic heart failure. The exceptional stability and control provided by TFX have the potential to enable substantial unloading of the right heart and hemodynamic normalization. In this study, the vast majority of procedures were completed without the need to re-cross the heart or use a stiff guidewire, and the data demonstrated an excellent safety profile with marked improvements in right heart function and overall clinical recovery. Together, these findings underscore the promise of this technology for pulmonary embolism and cardiovascular disease.”
Trial results showed:
- No major adverse events, including major bleeding, within 48 hours
- 80% of procedures were completed without re-crossing the heart
- 70% of procedures were completed without use of a stiff guidewire
- 32% improvement in mean pulmonary artery pressure (mPAP), decreasing intraprocedurally from 30.8 mmHg to 20.8 mmHg
- 29% improvement in mean right ventricle/left ventricle (RV/LV) ratio, decreasing from 1.4 to 1.0 at 48 hours post-procedure
- 71% improvement in mean modified Medical Research Council (mMRC) Dyspnea Scale at 48 hours (2.4 points) and 94% improvement at 30 days (3.2 points)
“SPIRARE I provides encouraging early evidence that improved procedural stability and control enabled by TFX can translate into meaningful improvements in cardiac function and patient outcomes,” said Charles Love, Chief Science Officer of Jupiter Endovascular. “Pulmonary embolism is fundamentally a cardiac disease, yet up to one-third of patients remain undertreated and continue to experience persistent symptoms and hemodynamic impairment that may contribute to right heart failure. We believe cardiac function and clinical recovery are the best measures of treatment efficacy in PE, and it is gratifying to see the therapeutic potential of TFX reflected in the SPIRARE I results through this cardiac-centric lens. We are deeply grateful to our pioneering physician partners for their collaboration, and above all, to the patients who placed their trust in us. We look forward to building upon these results in our ongoing SPIRARE II pivotal trial in the U.S. and Europe.”
To learn more, visit www.jupiterendo.com.


 October 31, 2025
October 31, 2025 









