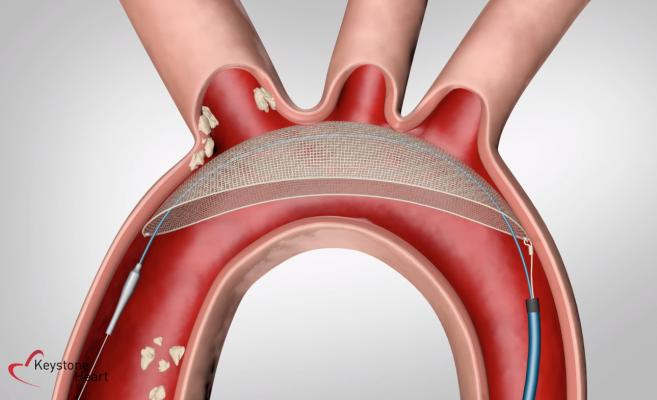
The Keystone Heart TriGuard 3 (TG3) is a self-stabilizing cerebral embolic deflection filter used in patients undergoing transcatheter aortic valve replacement (TAVR). The device is designed to reduce TAVR complications of cerebral embolization and ischemic stroke.
October 15, 2020 – The REFLECT II randomized clinical trial evaluating the safety and efficacy of the Keystone Heart TriGuard 3 (TG3), a self-stabilizing cerebral embolic deflection filter used in patients undergoing transcatheter aortic valve replacement (TAVR) met the primary safety endpoint, but did not demonstrate superiority over TAVR without use of embolic protection. The device is designed to reduce TAVR complications of cerebral embolization and ischemic stroke.
Findings were presented today at the Transcatheter Cardiovascular Therapeutics (TCT) Connect 2020 virtual conference.
A key measure for superiority was based on use of diffusion-weighted magnetic resonance imaging (DWI-MRI) to detect ischemic brain lesions caused by emboli post-procedure. The measure of total volume of cerebral lesions (TLV) was used and the median TLV was no different with TG3 protection compared to patients who did not have the device.
“Compared to controls, the primary 30-day safety endpoint was higher with TriGuard 3 due primarily to TAVR related vascular and bleeding complications,” said Jeffrey W. Moses, M.D. He is a professor of cardiology at Columbia University Vagelos College of Physicians and Surgeons, director of interventional cardiovascular therapeutics, NewYork-Presbyterian/Columbia University Irving Medical Center and director of advanced cardiac interventions, St. Francis Hospital and Heart Center. “While the
study did not demonstrate superiority of TriGuard 3 compared to pooled controls for the primary hierarchical efficacy endpoint, a post hoc DW-MRI analysis suggests that TG3 may reduce larger ischemic lesions. Improved device stability to achieve reliable, complete cerebral coverage may improve outcomes.”
REFLECT II intended to randomize 295 patients 2:1 to TAVR with TG3 vs. control. The primary safety endpoint was a composite of all-cause mortality, stroke, life-threatening or disabling bleeding, stage 2/3 acute kidney injury, coronary artery obstruction requiring intervention, major vascular complication, and valve-related dysfunction requiring intervention (VARC 2 defined) at 30 days. The endpoint was compared with a performance goal (PG) of 34.4%. The primary efficacy endpoint was a hierarchical composite of all-cause mortality or stroke at 30 days, NIHSS worsening, absence of DWI MRI lesions post-procedure, and TLV by DWI MRI. Cumulative scores derived by the Finkelstein-Schoenfeld method were summed for each patient and compared between groups.
The REFLECT II analysis population included 283 patients [41 roll-in, 121 randomized to TG3 and 121 controls (58 randomized in phase II and 63 pooled from REFLECT phase I)]. TG3 was delivered and positioned in the aortic arch prior to TAVR in 100% of cases and retrieved intact in all cases.
After enrollment of 179 of the 225 planned randomized patients, the sponsor suspended trial enrollment with the concurrence of the FDA and DMC. After limited unblinding and review of the data, Keystone Heart decided to formally close the study and proceed with the 510(k) marketing application.
TG3 met the primary safety endpoint (22.5% vs 34.4% PG, p non-inferiority=0.0001). However, superiority for the primary efficacy endpoint was not met, with similar win-ratios and win% (TG3 0.84 (45.7%) vs 1.19 (54.3%), p=0.857) between groups. Median TLV was not different with TG3 protection (215.39 mm3 vs 188.09 mm3, p=0.405).
The REFLECT II trial was funded by Keystone Heart. Dr. Moses reported no disclosures.
For more information: www.tctconference.com
Find additional TCT 2020 news, video and late-breaking studies


 May 06, 2026
May 06, 2026 









